Coating titania nanoparticles with carbon could result in a safer UV filter to be used in sunscreen, say Italian scientists.
Long-term exposure to UVA and UVB radiation from sunlight can cause wrinkles, damaged skin and, in some cases, skin cancer. Titania (TiO2), one of the main components in sunscreens, can absorb and scatter UVA and UVB radiation. However, titania can also become reactive under UV rays and in contact with water, generating free radicals that cause skin damage.
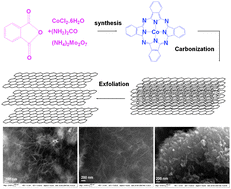
Now, Ivana Fenoglio from the University of Torino and Stefano Livraghi from the Institute for Health and Consumer Protection, Ispra, and their teams have modified the surface of titania nanoparticles to decrease their reactivity under UV. The groups coated the nanoparticles with ethylene glycol and heated the resulting compound to 300ºC to carbonise it. They found that this reduced the nanoparticles’ oxidative power and consequently decreased free radical formation. ‘It was very surprising to find out that by using ethylene glycol as a precursor, the formation of free radicals is reduced. This differs from the results of other studies done with titania nanoparticles modified with carbon,’ says Fenoglio.

Long-term exposure to UV radiation from sunlight can cause wrinkles, damaged skin and, in some cases, skin cancer
‘To use titania particles for skin care, a delicate balance is needed to prevent formation of reactive oxygen species, which have been suspected to cause skin damage, without affecting the desirable optical properties,’ says Sefik Suzer, an expert in inorganic nanoparticles at Bilkent University in Ankara, Turkey. ‘This research will undoubtedly help in developing a new generation of cosmetic products as well as leading to formulation of new routes for special applications of titania.’
‘This research may be a starting point for setting up protocols to produce UV filters that may find applications in different fields including the cosmetics industry,’ says Fenoglio.
Lorena Tomas Laudo
Download the full ChemComm communication to find out more about this work.
Comments Off on Safer sunscreens