We are delighted to share with you the top 25 most downloaded articles in Chemical Science from April–June 2014.

Novel hole transporting materials based on triptycene core for high efficiency mesoscopic perovskite solar cells
Anurag Krishna, Dharani Sabba, Hairong Li, Jun Yin, Pablo P. Boix, Cesare Soci, Subodh G. Mhaisalkar and Andrew C. Grimsdale
DOI: 10.1039/C4SC00814F
Copper catalyzed Heck-like cyclizations of oxime esters
Adele Faulkner, Nicholas J. Race, James S. Scott and John F. Bower
DOI: 10.1039/C4SC00652F
Enantioselective direct α-alkylation of cyclic ketones by means of photo-organocatalysis
Elena Arceo, Ana Bahamonde, Giulia Bergonzini and Paolo Melchiorre
DOI: 10.1039/C4SC00315B
Oxygen nucleophiles as reaction partners in photoinduced, copper-catalyzed cross-couplings: O-arylations of phenols at room temperature
Yichen Tan, José María Muñoz-Molina, Gregory C. Fu and Jonas C. Peters
DOI: 10.1039/C4SC00368C
Efficient C–H bond activations via O2 cleavage by a dianionic cobalt(II) complex
Andy I. Nguyen, Ryan G. Hadt, Edward I. Solomon and T. Don Tilley
DOI: 10.1039/C4SC00108G
Sunlight photocatalyzed regioselective β-alkylation and acylation of cyclopentanones
Megumi Okada, Takahide Fukuyama, Keiichi Yamada, Ilhyong Ryu, Davide Ravelli and Maurizio Fagnoni
DOI: 10.1039/C4SC01072H
Copper catalyzed direct alkenylation of simple alkanes with styrenes
Yefeng Zhu and Yunyang Wei
DOI: 10.1039/C4SC00093E
Transition metal-catalyzed direct nucleophilic addition of C–H bonds to carbon–heteroatom double bonds
Xi-Sha Zhang, Kang Chen and Zhang-Jie Shi
DOI: 10.1039/C3SC53115E
Evaluating metal–organic frameworks for natural gas storage
Jarad A. Mason, Mike Veenstra and Jeffrey R. Long
DOI: 10.1039/C3SC52633J
Selective radical amination of aldehydic C(sp2)–H bonds with fluoroaryl azides via Co(II)-based metalloradical catalysis: synthesis of N-fluoroaryl amides from aldehydes under neutral and nonoxidative conditions
Li-Mei Jin, Hongjian Lu, Yuan Cui, Christopher L. Lizardi, Thiago N. Arzua, Lukasz Wojtas, Xin Cui and X. Peter Zhang
DOI: 10.1039/C4SC00697F
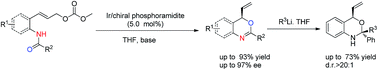
Catalytic enantioselective synthesis of 2-aryl-chromenes
Bi-Shun Zeng, Xinyi Yu, Paul W. Siu and Karl A. Scheidt
DOI: 10.1039/C4SC00423J
Dialkylbiaryl phosphines in Pd-catalyzed amination: a user’s guide
David S. Surry and Stephen L. Buchwald
DOI: 10.1039/C0SC00331J
Frustrated Lewis pair chemistry of carbon, nitrogen and sulfur oxides
Douglas W. Stephan and Gerhard Erker
DOI: 10.1039/C4SC00395K
Carbon–fluorine bond cleavage in fluoroarenes via a niobium(III) imido complex: from stoichiometric to catalytic hydrodefluorination
Thomas L. Gianetti, Robert G. Bergman and John Arnold
DOI: 10.1039/C4SC00006D
From themed collection Celebrating the 2014 RSC Prize and Award Winners
Rh(III)-catalyzed C–H functionalization/aromatization cascade with 1,3-dienes: a redox-neutral and regioselective access to isoquinolines
Dongbing Zhao, Fabian Lied and Frank Glorius
DOI: 10.1039/C4SC00628C
Iodoarene-catalyzed fluorination and aminofluorination by an Ar-I/HF·pyridine/mCPBA system
Satoru Suzuki, Tomohiro Kamo, Kazunobu Fukushi, Takaaki Hiramatsu, Etsuko Tokunaga, Toshifumi Dohi, Yasuyuki Kita and Norio Shibata
DOI: 10.1039/C3SC53107D
Complete stereodivergence in the synthesis of 2-amino-1,3-diols from allenes
Christopher S. Adams, R. David Grigg and Jennifer M. Schomaker
DOI: 10.1039/C4SC01214C
An ExBox [2]catenane
Michal Juríček, Jonathan C. Barnes, Nathan L. Strutt, Nicolaas A. Vermeulen, Kala C. Ghooray, Edward J. Dale, Paul R. McGonigal, Anthea K. Blackburn, Alyssa-Jennifer Avestro and J. Fraser Stoddart
DOI: 10.1039/C4SC00488D
From themed collection Celebrating the 2014 RSC Prize and Award Winners
Aluminium–ligand cooperation promotes selective dehydrogenation of formic acid to H2 and CO2
T. W. Myers and L. A. Berben
DOI: 10.1039/C4SC01035C
Synthesis, electronic structure and reactivity of bis(imino)pyridine iron carbene complexes: evidence for a carbene radical
Sarah K. Russell, Jordan M. Hoyt, Suzanne C. Bart, Carsten Milsmann, S. Chantal E. Stieber, Scott P. Semproni, Serena DeBeer and Paul J. Chirik
DOI: 10.1039/C3SC52450G
Non-directed allylic C–H acetoxylation in the presence of Lewis basic heterocycles
Hasnain A. Malik, Buck L. H. Taylor, John R. Kerrigan, Jonathan E. Grob, K. N. Houk, J. Du Bois, Lawrence G. Hamann and Andrew W. Patterson
DOI: 10.1039/C3SC53414F
Rethinking the term “pi-stacking”
Chelsea R. Martinez and Brent L. Iverson
DOI: 10.1039/C2SC20045G
From themed collection Physical Chemistry
Stereoselective allylboration of imines and indoles under mild conditions. An in situ E/Z isomerization of imines by allylboroxines
Rauful Alam, Arindam Das, Genping Huang, Lars Eriksson, Fahmi Himo and Kálmán J. Szabó
DOI: 10.1039/C4SC00415A
Cobaltate anion couples terminal dienes with trifluoroacetic anhydride: a direct fluoroacylation of 1,3-dienes
Benjamin L. Kohn and Tomislav Rovis
DOI: 10.1039/C4SC00743C
Phosphorescent nematic hydrogels and chromonic mesophases driven by intra- and intermolecular interactions of bridged dinuclear cyclometalated platinum(II) complexes
Xin-Shan Xiao, Wei Lu and Chi-Ming Che
DOI: 10.1039/C4SC00143E
Chemical Science is the Royal Society of Chemistry’s flagship journal; publishing research articles of exceptional significance and high-impact reviews from across the chemical sciences. The journal’s latest (2013) Impact Factor is 8.6. Research in Chemical Science is not only of the highest quality but also has excellent visibility; this is reflected in our latest citation profile.
Chemical Science is moving to Gold Open Access from Issue 1, 2015. It will be the world’s first high-quality Open Access chemistry journal.
Submit your exceptional research to Chemical Science today!
Stay up to date with Chemical Science
Be among the first to hear about the newest articles being published – Sign-up to our journal news alert to receive information about most read articles, themed issues, journal news, as well as calls for papers and invitations.











![The binding of calix[4]pyrrole to guests immobolised on a surface is, kinetically, slower than in bulk solution](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C4SC01745E)
 A fluorinated analogue of the naturally occurring aminoglycoside neomycin – well known as an over-the-counter ointment for minor skin abrasions – could lead to
A fluorinated analogue of the naturally occurring aminoglycoside neomycin – well known as an over-the-counter ointment for minor skin abrasions – could lead to