Proton exchange membranes (PEMs) are essential to the functionality of fuel cells. They conduct protons in electrolytes and drive electricity generation by oxidizing fuels. Following the success of Nafion® –– a family of commercial proton-conductive fluoropolymers –– materials researchers around the globe are developing innovative PEMs with high proton conductivities and affordable prices.
A group of Japanese researchers has recently synthesized self-standing polymer films with a gyroid nanostructure. These films possess two unique characteristics that other PEMs rarely have: a high proton conductivity in the order of 10-1 S/cm and retention of the conductivity across a wide temperature range (20-120 °C). This finding has been published in Chem. Sci. (doi: 10.1039/C9SC00131J).
The authors used a tailor-made macromolecule, Diene-GZI (Figure 1a), as the building block. It had an amphipathic structure, with one end being a hydrophilic zwitterionic group and another end of a hydrophobic alkyl chain. When mixed with bis(trifluoromethanesulfonyl)imide and water, multiple Diene-GZI molecules could assemble together into a gyroid network –– an infinitely periodic minimal surface (Figure 1b). After the self-assembly, ultra-violet-irradiation-induced polymerization solidified the morphology of the gyroid nanostructure.
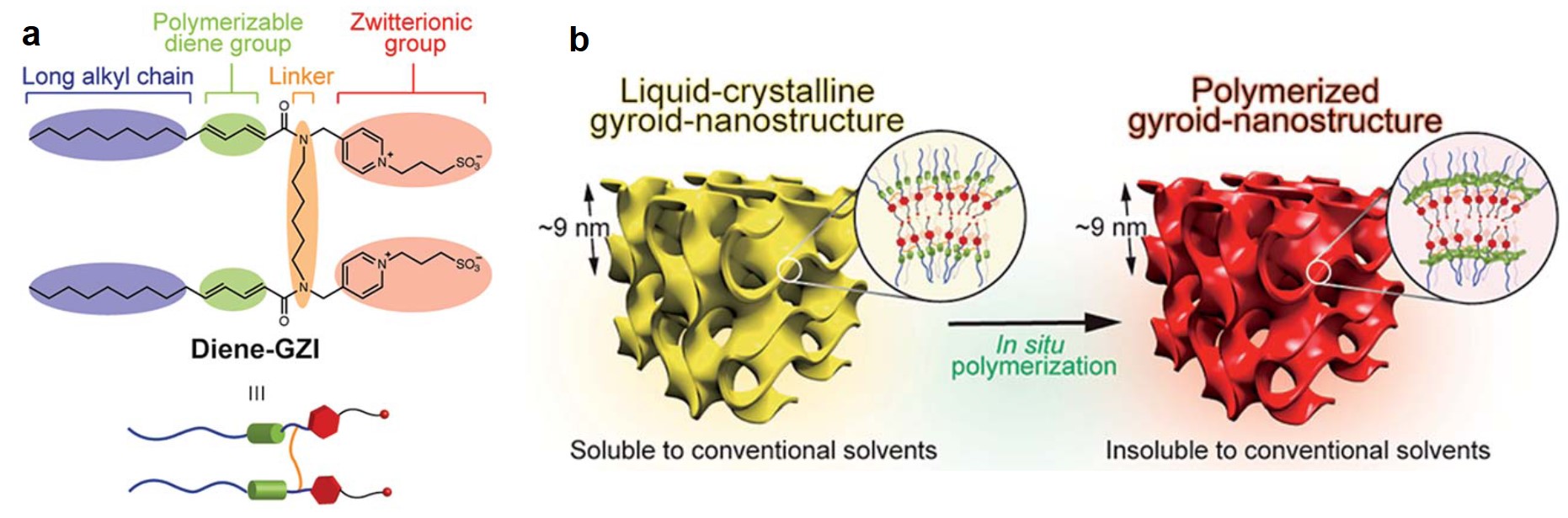
Figure 1. (a) The molecular structure of Diene-GZI. (b) Solidification of the self-assembled gyroid via polymerization.
The high proton conductivity of the polymer film originated from its three-dimensional gyroid structure. Since the gyroid surface was densely coated with the hydrophilic zwitterionic chains, the film could readily uptake as high as 15.6 wt.% of water at a relative humidity of 90%. The adsorbed water layers formed a three-dimensional continuous pathway along the gyroid surface, serving as proton-conduction expressways and resulting in a high conductivity in the order of 10-1 S/cm. Due to the strong binding force between water and the zwitterionic groups, heating the polymer film to 120 °C did not decrease the water content significantly, and thus, the proton conductivity remained high. Additionally, the control films with no gyroid structures were unable to compete with the gyroid film in terms of proton conductivities within the measured temperature range (Figure 2).
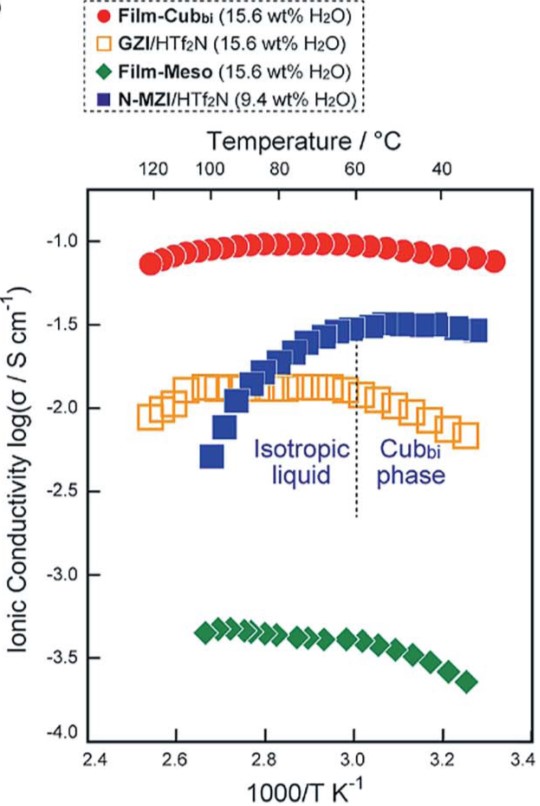
Figure 2. The dependence between proton conductivities and temperature. Legends: red solid circles – gyroid film; others – control samples without the gyroid nanostructure.
This work highlights the critical role of rational design of raw materials to augment the proton conductivities of PEMs. The advantage of the gyroid phase in speeding up ion diffusion could also inspire innovative materials in applications demanding ultrafast ion transport, e.g., supercapacitor electrodes.
To find out more, please read:
Gyroid Structured Aqua-Sheets with Sub-Nanometer Thickness Enabling 3D Fast Proton Relay Conduction
Tsubasa Kobayashi, Ya-xin Li, Ayaka Ono, Xiang-bing Zeng, and Takahiro Ichikawa
Chem. Sci., 2019, 10, 6245-6253
About the blogger:
 Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.
Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.