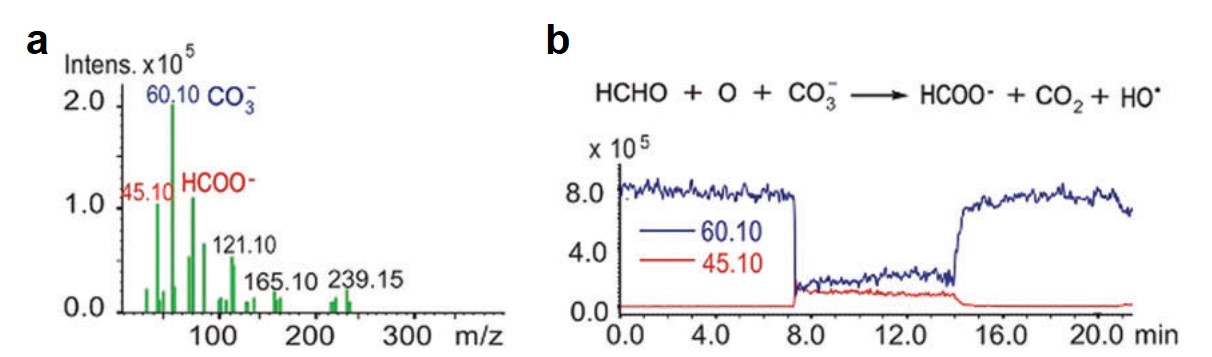
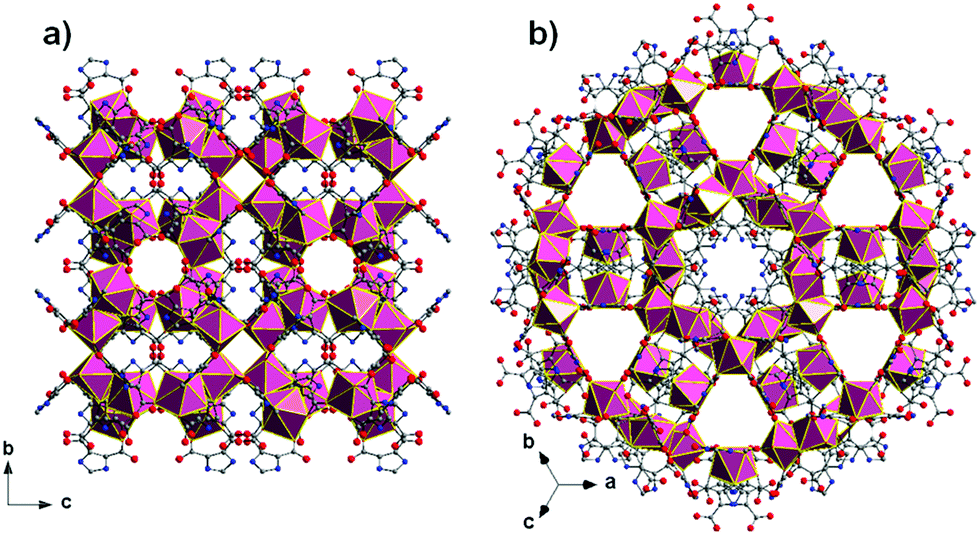
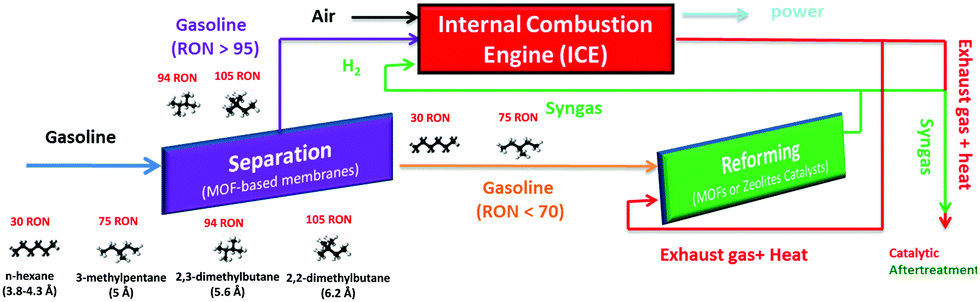
All of the referee-recommended articles below are free to access until 4th November 2018.
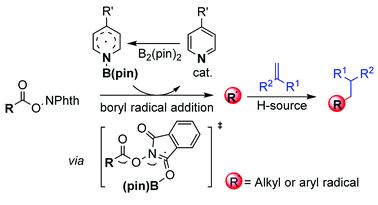
Organocatalytic decarboxylative alkylation of N-hydroxy-phthalimide esters enabled by pyridine-boryl radicals
Liuzhou Gao, Guoqiang Wang, Jia Cao, Dandan Yuan, Cheng Xu, Xuewen Guo and Shuhua Li
Chem. Commun., 2018, Advance Article
DOI: 10.1039/C8CC06152A, Communication
_______________________________________________________________________________
A new C,N-cyclometalated osmium(II) arene anticancer scaffold with a handle for functionalization and antioxidative properties
Enrique Ortega, Jyoti G. Yellol, Matthias Rothemund, Francisco J. Ballester, Venancio Rodríguez, Gorakh Yellol, Christoph Janiak, Rainer Schobert and José Ruiz
Chem. Commun., 2018,54, 11120-11123
DOI: 10.1039/C8CC06427J, Communication
_______________________________________________________________________________
Descriptors of magnetic anisotropy revisited
Mauro Perfetti and Jesper Bendix
Chem. Commun., 2018, Advance Article
DOI: 10.1039/C8CC05756G, Communication
_______________________________________________________________________________
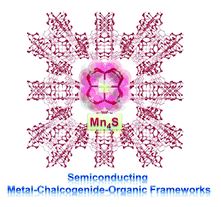
A semiconducting metal-chalcogenide–organic framework with square-planar tetra-coordinated sulfur
Huajun Yang, Min Luo, Zhou Wu, Wei Wang, Chaozhuang Xue and Tao Wu
Chem. Commun., 2018,54, 11272-11275
DOI: 10.1039/C8CC06997B, Communication
_______________________________________________________________________________
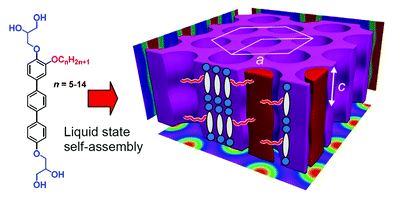
Transition between tangential and co-axial liquid crystalline honeycombs in the self-assembly of Y-shaped bolapolyphiles
Anne Lehmann, Marko Prehm, Changlong Chen, Feng Liu, Xiangbing Zeng, Goran Ungar and Carsten Tschierske
Chem. Commun., 2018, Advance Article
DOI: 10.1039/C8CC06281A, Communication
_______________________________________________________________________________
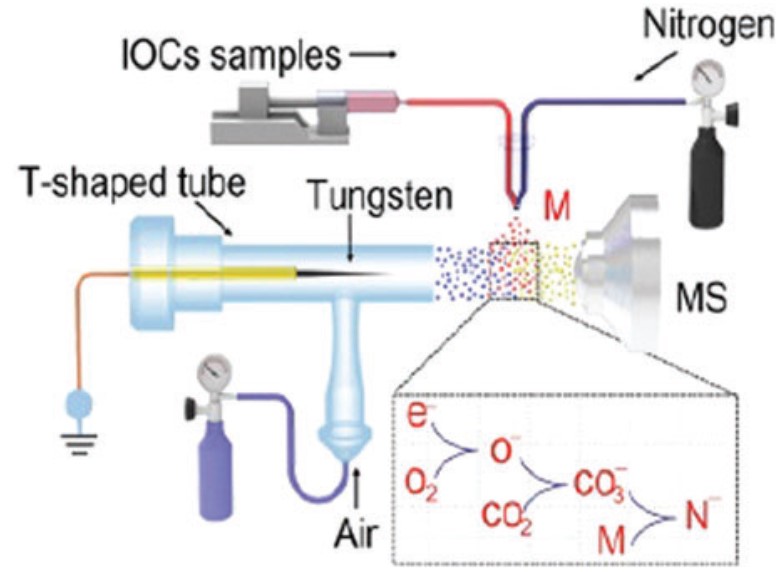
Unraveling the isomeric heterogeneity of glycans: ion mobility separations in structures for lossless ion manipulations
Gabe Nagy, Isaac K. Attah, Sandilya V. B. Garimella, Keqi Tang, Yehia M. Ibrahim, Erin S. Baker and Richard D. Smith
Chem. Commun., 2018, Advance Article
DOI: 10.1039/C8CC06966B, Communication














 Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at
Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at 









![[double bond, length as m-dash]](https://www.rsc.org/images/entities/h2_char_e001.gif) Ge
Ge![[double bond splayed right]](https://www.rsc.org/images/entities/h2_char_e00a.gif) double bond
double bond


 About the author
About the author