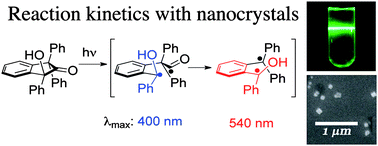
US scientists have demonstrated for the first time that a metal–carbon multiple bond complex can activate methane. The work could open the way for efficiently converting methane into useful hydrocarbons including ethylene, one of the fundamental building blocks of the chemical industry.
Methane is the principal component of natural gas and a major contributor to global warming. It is therefore desirable to find cleaner and cheaper ways of using it as a resource by converting it into more useful chemicals. However, such reactions pose significant challenges because of methane’s low reactivity and strong tetrahedral C–H bonds, which are not readily accessible for chemical attack and activation.
The research could ultimately enable scientists to use methane as a feedstock for C–C bond formation, important for the production of chemicals including pharmaceuticals and plastics, say Daniel Mindiola (Indiana University) and colleagues.
Download Mindiola’s Edge article to find out more.












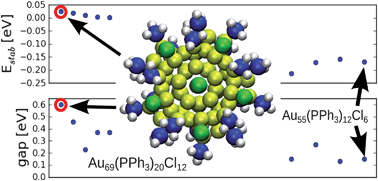 Teams from Germany, the US and Finland have re-investigated the structure of a famous gold-phosphine-halide compound.
Teams from Germany, the US and Finland have re-investigated the structure of a famous gold-phosphine-halide compound. They visualised clustering of lipid molecules in nanodomains with a spatial resolution one order of magnitude better than the diffraction limit and were able to resolve the detailed structure of the domains.
They visualised clustering of lipid molecules in nanodomains with a spatial resolution one order of magnitude better than the diffraction limit and were able to resolve the detailed structure of the domains.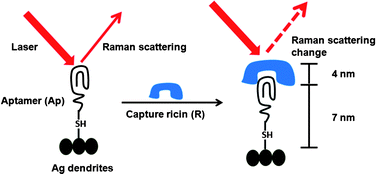 Ricin is a protein toxin naturally present in the castor bean plant (Ricinus communis); it is classified as a ‘select’ bioterror agent and was involved in a recent bioterrorism attack plot targeting US hotels and restaurants at multiple locations, as reported by the Department of Homeland Security officials in December 2010.
Ricin is a protein toxin naturally present in the castor bean plant (Ricinus communis); it is classified as a ‘select’ bioterror agent and was involved in a recent bioterrorism attack plot targeting US hotels and restaurants at multiple locations, as reported by the Department of Homeland Security officials in December 2010.