Chemical Science Reviewer Spotlight – June 2025
To further thank and recognise the support from our excellent reviewer community, we are highlighting reviewers who have provided exceptional support to the journal over the past year.
This month, we’ll be highlighting Dr Stephen Fielden, Dr Franziska Thomas, Professor Luca Dell’Amico and Professor Xin-Yao Yu. We asked our reviewers a few questions about what they enjoy about reviewing, and their thoughts on how to provide a useful review.

Dr Stephen Fielden, University of Birmingham. I combine supramolecular chemistry and polymer science to develop new nanotechnology. I am particularly interested in developing complex systems of nanoparticles that influence and control each other.

Dr Franziska Thomas, Heidelberg University. My research focuses on designing and synthesising functional miniproteins — peptides with a defined three-dimensional structure that can contain natural and non-natural building blocks.

Professor Luca Dell’Amico, University of Padova. We build molecules using the most sustainable methods available or developing new ones. We harness the transformative power of light, either by directly exciting the molecules or by employing organic photocatalysts. We are fascinated by new reaction manifolds that open up novel pathways to access previously unattainable scaffolds and bioactive molecules. We study these new synthetic transformations in depth using a range of optical and spectroscopic techniques, as well as computational methods.

Professor Xin-Yao Yu, Anhui University. My research centers on the design and synthesis of advanced materials for energy storage and conversion.
What encouraged you to review for Chemical Science?
Dr Stephen Fielden: I know that a paper considered for Chemical Science is going to contain exciting new results and provide a scientific advance that I haven’t seen before. Also, I like to help the RSC to continue publishing impactful work in an open access format.
Dr Franziska Thomas: Chemical Science is an interdisciplinary journal that I regularly read and in which I have also published articles. As my own manuscripts have been reviewed by other scientists, I consider it my duty to review manuscripts in return.
Professor Luca Dell’Amico: Chemical Science is a go-to journal for us chemists. It always maintains excellent research standards, which motivates me to do my best as a reviewer and contribute to its continued growth.
Professor Xin-Yao Yu: I was motivated by the journal’s reputation for publishing high-impact, interdisciplinary chemical research, coupled with its commitment to rigorous yet constructive peer review. As a researcher in materials chemistry, I see reviewing as a way to contribute to maintaining scientific standards while engaging with cutting-edge work in my field.
What do you enjoy most about reviewing?
Dr Stephen Fielden: I really enjoy being one of the first people to see new science. In addition, it’s satisfying to know that I can help ensure that new advances are impactful as possible.
Dr Franziska Thomas: It is always nice to read about new and refreshing concepts. What I enjoy most is offering my colleagues suggestions on how to improve a manuscript, thereby enhancing the research it describes.
Professor Luca Dell’Amico: It’s when I see exciting new findings and the authors’ enthusiasm, combined with a detailed and precise analysis of new reaction manifolds and/or applications, that I feel truly inspired.
Professor Xin-Yao Yu: The most rewarding aspect of reviewing is the opportunity to engage with innovative research at the forefront of the field. I enjoy critically evaluating scientific arguments, identifying strengths that deserve recognition, and providing constructive feedback to help authors refine their work. Additionally, reviewing allows me to learn from diverse research approaches and gain fresh perspectives that often inspire new ideas in my own work.
What are you looking for in a paper that you can recommend for acceptance in Chemical Science?
Dr Stephen Fielden: I am looking for a new concept that draws together disparate areas of research. If the science has been done well to evidence the claims behind this new concept then I am happy.
What makes a paper truly stand out for you when reviewing a paper?
Dr Franziska Thomas: An exceptional manuscript is characterised by an innovative research idea that is systematically implemented through a series of well-founded experiments. Additionally, an outstanding publication is supported by figures and schemes that convey the research results clearly, even when the text is not read.
Professor Luca Dell’Amico: As mentioned above, I believe it’s a combination of the authors’ enthusiasm in presenting their findings and the deep mechanistic understanding of a new process or mechanism with broader applications.
What single piece of advice would you give to someone about to write their first review?
Dr Franziska Thomas: Read the manuscript through once, then put it aside for a day before reading it again in detail. Don’t forget to read the supplementary material.
How has your approach to peer reviewing changed over time?
Professor Luca Dell’Amico: I always follow the same approach: first, a general read-through, and then a more detailed understanding of the work by going section by section, also analyzing the supporting materials. Sometimes we’re overwhelmed with review requests, but I always stick to this method—perhaps being a bit late at times—in order to maintain the highest possible standards. We owe this to both the authors (as fellow researchers) and to the journal.
What advice would you give a first-time author looking to maximise their chances of successful peer review?
Professor Xin-Yao Yu: Clarify the scientific significance; clearly articulate how the work addresses a critical challenge in the field and why it matters beyond the specific methodology. Prioritize logical flow; organize the manuscript with a strong narrative structure, ensuring each section (introduction, methods, results, discussion) builds coherently toward the central conclusion. Anticipate reviewer concerns; proactively address potential limitations, compare your work with existing literature, and provide robust data to support claims. Pay attention to presentation; use high-quality figures and concise language to enhance readability.
What has been your biggest learning point from reviewing?
Dr Stephen Fielden: Insight into the peer review process is essential for scientists to learn their craft and to see that every good research paper starts as a ‘rough diamond’.
Did reviewing for Chemical Science affect how you approached preparation of your recent publication with us?
Professor Xin-Yao Yu: Yes, the reviewing experience has significantly influenced my approach to manuscript preparation for Chemical Science. Through evaluating other researchers’ work, I gained a deeper understanding of the journal’s expectations for novelty, rigor, and interdisciplinary appeal. This perspective helped me refine the narrative of our recent submission, ensuring we emphasized the broader implications of our findings and proactively addressed potential cross-disciplinary questions. Additionally, reviewing highlighted the importance of clear data presentation and robust supporting information, which we prioritized in our manuscript to enhance transparency and scientific integrity.
Tune in soon to meet our next group of #ChemSciReviewers!
If you want to learn more about how we support our reviewers, check out our Reviewer Hub.
Interested in joining our ever-growing reviewer community? Apply here now!
Comments Off on Chemical Science Reviewer Spotlight – June 2025





































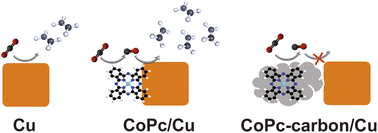



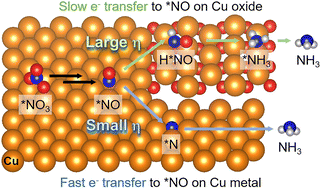









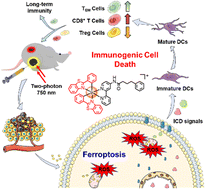













![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C isomers
C isomers![Graphical abstract: High-throughput single-cell mass spectrometry enables metabolic network analysis by resolving phospholipid C [[double bond, length as m-dash]] C isomers](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3SC06573A&imageInfo.ImageIdentifier.Year=2024)




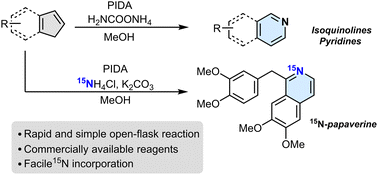
![Graphical abstract: 1,2-Disubstituted bicyclo[2.1.1]hexanes as saturated bioisosteres of ortho-substituted benzene](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3SC05121H&imageInfo.ImageIdentifier.Year=2023)
![Graphical abstract: Synthesis of polysubstituted bicyclo[2.1.1]hexanes enabling access to new chemical space](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3SC03083K&imageInfo.ImageIdentifier.Year=2023)


![Graphical abstract: Lemniscular carbon nanohoops with contiguous conjugation from planar chiral [2.2]paracyclophane: influence of the regioselective synthesis on topological chirality](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D2SC06825G&imageInfo.ImageIdentifier.Year=2023)

![Graphical abstract: 2,5-disubstituted bicyclo[2.1.1]hexanes as rigidified cyclopentane variants](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3SC02695G&imageInfo.ImageIdentifier.Year=2023)

![Graphical abstract: Lewis acid catalyzed [4+2] annulation of bicyclobutanes with dienol ethers for the synthesis of bicyclo[4.1.1]octanes](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4SC02767A&imageInfo.ImageIdentifier.Year=2024)
![Graphical abstract: Palladium-catalyzed decarboxylative (4 + 3) cycloadditions of bicyclobutanes with 2-alkylidenetrimethylene carbonates for the synthesis of 2-oxabicyclo[4.1.1]octanes](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D4SC02998D&imageInfo.ImageIdentifier.Year=2024)

![Graphical abstract: De novo synthesis of inherently chiral heteracalix[4]aromatics from enantioselective macrocyclization enabled by chiral phosphoric acid-catalyzed intramolecular SNAr reaction](https://pubs.rsc.org/en/Image/Get?imageInfo.ImageType=GA&imageInfo.ImageIdentifier.ManuscriptID=D3SC06436K&imageInfo.ImageIdentifier.Year=2024)














