I feel pretty safe saying that the development of effective antimicrobial drugs (looking at you penicillin!) was one of the most significant pharmacological events of recent history. Unfortunately, the widespread and often indiscriminate use of antibiotics has created an environment where bacteria with evolved drug resistances, colloquially known as “superbugs,” pose a serious threat to global health. While development of new small-molecule antimicrobial drugs is still ongoing, scientists are exploring alternative approaches as well. Of interest are antimicrobial peptides, found in plants and animals as a part of native immune systems. The most common are cationic antimicrobial peptides (CAMPs) comprised of 10-50 amino acids that possess both cationic and hydrophobic subunits and an overall positive charge. This allows them to strongly electrostatically interact with negatively charged bacterial cell membranes without disrupting neutral animal cells. Unfortunately, CAMPs have proved challenging to commercialize with generally unfavorable activities and issues with large-scale manufacture.
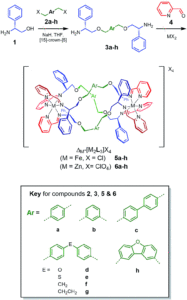
Researchers in the UK and Czech Republic recently developed a range of cationic metallohelices (Figure 1) that demonstrate structure-dependent activity against both Gram-positive and -negative bacteria. The diamine ligands form cleanly in the presence of [15]-crown-[5] and upon combination with 2-pyridinecarboxaldehyde and a metal salt self-assemble into the iron or zinc metallohelices. The metallohelices consist of a single enantiomer, as the ligands are optically pure, as determined by NMR and single-crystal X-ray diffraction when possible. Altering the aryl linker unit in the ligand the overall size and shape of the metallohelix in both zinc and iron derivatives. The iron metallohelices are water compatible, with lifetimes exceeding 10 days even in highly acidic conditions, and thus suitable for antimicrobial activity screening.
The researchers used in vitro studies to find minimum inhibitory concentrations (MICs), the lowest concentration necessary to see bacterial growth inhibition. While the metallohelices demonstrate the highest activity towards Gram-positive bacteria, some showed lethal effects on Gram-negative E. coli in 20-40 minutes. The 5b helices with a para-benzene bridging group acted most selectively on E. coli with the Λ-5b enantiomer acting as the champion compound and selected for further mechanistic study. The researchers exposed a notorious E. coli strain to inhibitory levels of Λ-5b in an attempt to isolate resistant mutants. The 17 isolates showed only slight tolerance increases rather than true resistance and could be classified into 4 genetic sub-types. Two of the sub-types developed mutations that altered the biophysical properties of their outer membrane, a third lost the ability to produce the vitamin B12 transporter protein, and the fourth lost the pO157 virulence plasmid (which makes this particularly E. coli strain particularly unpleasant). Taken in concert, these 4 sub-types show that tolerance can be derived from disrupting the ability of Λ-5b to interact with and cross the cellular membrane.

Figure 2. Fluorescence images of cells treated with Λ-5b coupled to a fluorescent probe, with arrows pointing to the localization of Λ-5b.
Given data suggesting the ability of Λ-5b to cross the cellular membrane, despite the relatively large of the metallohelices, the researchers used fluorescence microscopy to probe Λ-5b localization by coupling Λ-5b with a fluorescent label. The labeled metallohelix preferentially localized to regions in growing cells that contain anionic phospholipids (Figure 2). This indicates that Λ-5b can cross the cellular membrane and acts internally to the cell rather than simply acting via electrostatic interaction that disrupts the membrane. Overall, this work provides an exciting approach to developing novel anti-microbial drugs that mimic CAMPs with higher stability, activity, and easier synthesis.
To find out more please read:
Metallohelices that kill Gram-negative pathogens using intracellular antimicrobial peptide pathways
Daniel H. Simpson, Alexia Hapeshi, Nicola J. Rogers, Viktor Brabec, Guy J. Clarkson, David J. Fox, Ondrej Hrabina, Gemma L. Kay, Andrew K. King, Jaroslav Malina, Andrew D. Millard, John Moat, David I. Roper, Hualong Song, Nicholas R. Waterfield and Peter Scott
About the blogger:
 Beth Mundy is a PhD candidate in chemistry in the Cossairt lab at the University of Washington in Seattle, Washington. Her research focuses on developing new and better ways to synthesize nanomaterials for energy applications. She is often spotted knitting in seminars or with her nose in a good book. You can find her on Twitter at @BethMundySci.
Beth Mundy is a PhD candidate in chemistry in the Cossairt lab at the University of Washington in Seattle, Washington. Her research focuses on developing new and better ways to synthesize nanomaterials for energy applications. She is often spotted knitting in seminars or with her nose in a good book. You can find her on Twitter at @BethMundySci.