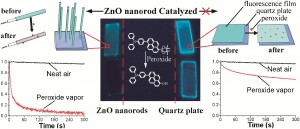
Gold nanoparticles capped with mercaptocarboxylic acids, followed by silver precipitation, have been used to develop latent fingerprints on paper as high quality negative images. Scientists writing in the journal ChemComm say that the effect stems from hydrogen bonding between the carboxylic group and the paper cellulose.
Recovering fingerprints from paper is a common task for forensic scientists, but often the developed marks are faint. A common approach, therefore, is to use a developing agent that sticks to the clean paper substrate, rather than the fingerprint itself, yielding a reversed image.
The technique described in this study is much less affected by sweat composition, and could improve the yield of latent fingerprints.
Read the ‘HOT’ ChemComm article today for free:
A novel approach to fingerprint visualization on paper using nanotechnology: reversing the appearance by tailoring the gold nanoparticles’ capping ligands
Sanaa Shenawi , Nimer Jaber , Joseph Almog and Daniel Mandler
Chem. Commun., 2013, DOI: 10.1039/C3CC41610K











 3D carbon nanotube forests are of particular interest in the electrochemical arenas of sensing and energy applications. Some researchers have suggested that it is necessary to use open-ended carbon nanotubes and carry out a pre-treatment or activation step to support fast electrochemistry, but is this always the case?
3D carbon nanotube forests are of particular interest in the electrochemical arenas of sensing and energy applications. Some researchers have suggested that it is necessary to use open-ended carbon nanotubes and carry out a pre-treatment or activation step to support fast electrochemistry, but is this always the case?