At a product launch in California last week Elon Musk introduced Tesla’s new electric semi-trailer truck. Musk sells a tantalising future: one where an electric fleet replaces vehicles which currently rely on fossil fuels. Central to this fleet are powerful rechargeable batteries. Lithium-ion batteries are favoured for many current applications, such as portable electronic devices and the current offerings of full and hybrid vehicles. In coming years they are projected to be the technology of choice for the large-scale applications mentioned above and for storing power generated from intermittent renewable energy sources.
A limiting factor in the widespread roll-out of lithium batteries is that lithium is an expensive resource with low natural abundance. Sodium offers a possible alternative and has the obvious benefits of being both very cheap, and one of the most abundant elements in the earth’s crust. The electrode materials used in lithium batteries cannot be used to make the sodium variant because the sodium ion is larger (1.02 Å compared to 0.76 Å for lithium) and damages the crystalline materials optimised for lithium.
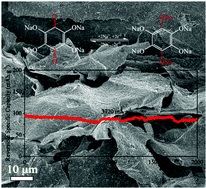
Researchers Gu, Gu and Yang at Beihang University in Beijing have reported the synthesis and performance of a novel anode material optimised for sodium. The material is a graphene-tetrahydroxybenzoquinone (Na4C6O6) hybrid, and is comprised of a porous graphene-oxide scaffold decorated with nanocrystals of Na4C6O6. Furthermore, X-ray photoelectron spectroscopy (XPS) reveals the homogenous distribution of sodium throughout this conducting material.
The electrochemical performance contrasts with previously reported materials of this type by exhibiting high cyclic stability. The reversible capacity of graphene-Na4C6O6 at a current density of 74.4 mA g-1 is 268 mA h g-1, a value which is steady over 60 cycles. This is competitive with the graphite anode materials found in lithium batteries, which have specific capacities between 200 and 400 mA h g-1. Furthermore the material performs well over a range of current densities, with reversible capacities of 95 – 211 mA h g-1 measured over a range of 3720 – 186 mA g-1.
With this work the authors contribute, at most, a viable candidate for the next rechargeable sodium battery and, at the very least, continued research into sustainable technologies. This ensures that in addressing our current energy challenges we are solving the problem, not delaying it.
To find out more please read:
3D organic Na4C6O6/graphene architecture for fast sodium storage with ultralong cycle life
Jianan Gu, Yue Gua and Shubin Yang
Chem. Commun., 2017, Advance Article
DOI: 10.1039/C7CC08045J, Communication
 About the author
About the author
Zoë Hearne is a PhD candidate in chemistry at McGill University in Montréal, Canada, under the supervision of Professor Chao-Jun Li. She hails from Canberra, Australia, where she completed her undergraduate degree. Her current research focuses on transition metal catalysis to effect novel transformations, and out of the lab she is an enthusiastic chemistry tutor and science communicator.