Nanomaterials are small and mighty. Scaling down to the molecular level imparts desirable properties to materials, for example high conductivity that is useful for electronic applications. Conjugated polymers (CPs) are one-dimensional nanomaterials with promising potential in optoelectronics and spintronics, due to their enhanced conductivity as a result of the delocalised π-electrons along the backbone of the polymer. Doubly-linked CPs, referred to as π-conjugated ladder polymers, have more unique features compared to their singly-linked counterparts, but such nanomaterials are often difficult to synthesise.
Polycyclic aromatic molecules such as acenes have shown potential as organic semiconductors and are therefore ideal materials for new CPs, but polymer formation by coupling the acenes is limited by repulsions between hydrogen atoms of adjacent acene motifs. These constraints can be overcome by installing wider ethynylene or cumulene π-conjugated bridges between the acenes, as demonstrated by researchers from Spain and the Czech Republic in their design and synthesis of a new one-dimensional π-conjugated ladder polymer (Figure 1). This ladder polymer is based on doubly-linked pentacenes (5 linearly-fused benzene rings).
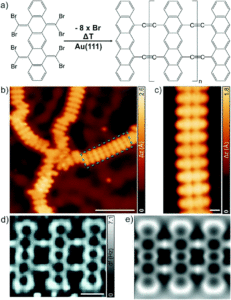
Figure 1. (a) The synthesis of the ladder polymer, starting from the 8BrPN precursor. (b) Wide STM image showing chains of the new polymer on the Au(111) surface. (c) Close-up high-resolution STM image of one polymer chain corresponding to the green rectangle in (b). (d) nc-AFM image of the molecular structure of the ladder polymer. (e) Simulated nc-AFM image of (d).
The new conjugated ladder polymer was synthesised by vacuum deposition of the precursor molecule (5,7,12,14-tetrakis(dibromomethylene)-5,7,12,14-tetrahydropentacene, 8BrPN) onto a gold surface, Au(111). Subsequent thermal treatment up to 360 °C resulted in the formation of ethynylene linkages between the pentacenes, forming a ladder polymer on the gold surface (Figure 1a). The researchers determined the ladder polymer structure using scanning tunnelling microscopy (STM) and non-contact atomic force microscopy (nc-AFM) techniques, as shown in Figure 1b-e. The STM images showed mostly one-dimensional chains with some disordered segments (Figure 1b), and the close-up image in Figure 1c confirms the double-linkages between each pentacene. The nc-AFM images show even more detail, confirming that the backbone of the polymer contains the pentacenes, doubly-linked to adjacent moieties in the 5,7,12 and 14 positions (Figure 1d/e). The nc-AFM characterisation also confirmed ethynylene (–C≡C–) rather than cumulene (=C=C=) linkages, shown by the brighter regions in the middle of the connections that correspond to the ethynylene triple bonds.
The researchers also probed the electronic structure of the new polymer on the gold surface using scanning tunnelling spectroscopy (STS). They determined an electronic bandgap that is 0.22 eV larger than that of a singly cumulene-linked pentacene polymer, thus demonstrating the advantages of the ladder-type structure in this instance. The technique used to create the pentacene ladder polymer in this report could be adopted for the creation of other π-conjugated ladder polymers with varying acenes, which could ultimately be useful for new nanomaterials in optoelectronics and spintronics.
To find out more, please read:
On-surface synthesis of doubly-linked one-dimensional pentacene ladder polymers
Kalyan Biswas, José I. Urgel, Ana Sánchez-Grande, Shayan Edalatmanesh, José Santos, Borja Cirera, Pingo Mutombo, Koen Lauwaet, Rodolfo Miranda, Pavel Jelínek,* Nazario Martín* and David Écija*
Chem. Commun., 2020, 56, 15309–15312
About the blogger:
 Dr. Samantha Apps recently finished her post as a Postdoctoral Research Associate in the Lu Lab at the University of Minnesota, USA, and obtained her PhD in 2019 from Imperial College London, UK. She has spent the last few years, both in her PhD and postdoc, researching synthetic nitrogen fixation and transition metal complexes that can activate and functionalise dinitrogen. Outside of the lab, you’ll likely find her baking at home, where her years of synthetic lab training has sparked a passion in kitchen chemistry too.
Dr. Samantha Apps recently finished her post as a Postdoctoral Research Associate in the Lu Lab at the University of Minnesota, USA, and obtained her PhD in 2019 from Imperial College London, UK. She has spent the last few years, both in her PhD and postdoc, researching synthetic nitrogen fixation and transition metal complexes that can activate and functionalise dinitrogen. Outside of the lab, you’ll likely find her baking at home, where her years of synthetic lab training has sparked a passion in kitchen chemistry too.










