Chemists use many rules to describe and predict molecular properties and behaviours. Hückel’s rule is a well-known example, and is used to determine the aromatic character of planar, cyclic molecules containing π-bonds. Aromaticity is the property that increases the stabilisation of a planar molecule by the delocalisation of electrons in π-bonds, and according to Hückel’s rule, a molecule is aromatic if it contains 4n + 2 π-electrons and antiaromatic if it contains 4n π-electrons. Whilst Hückel’s rule applies to the singlet ground state of a molecule (i.e. when all electrons are paired), Baird’s rule is used to establish aromaticity in the triplet state (i.e. two unpaired electrons), and is essentially the opposite; aromatic for 4n and antiaromatic for 4n + 2 π-electrons (Figure 1). Baird’s rule is useful for understanding the excited (triplet) state properties of molecules, but has so far been limited to describing organic species only.
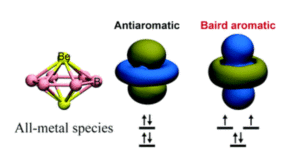
Figure 1. An example of a Baird aromatic all-metal species (left), with orbitals that exhibit Baird aromaticity in the triplet state (right), and Hückel antiaromaticity in the singlet state (centre)
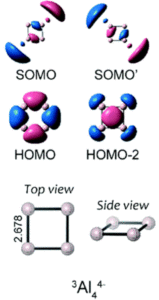
A collaborative effort by researchers in China, Spain and Poland have now demonstrated that Baird aromaticity can be translated to all-metal systems using a series of DFT experiments. The researchers selected derivatives of Al44-, a planar moiety with 4 π-electrons that is antiaromatic in the ground state (consistent with Hückel’s rule). They determined that in the triplet (T1) state, the two unpaired electrons in Al44- occupied σ molecular orbitals (MOs) instead of π MOs; these are the singly-occupied molecular orbitals (SOMOs) shown in Figure 2 that show anti-bonding character for the σ-radial system. The researchers could then infer a triple aromaticity for the triplet state of Al44- from the formal electron count: Hückel aromaticity of the 2 electrons in the σ-tangential system (HOMO); Hückel aromaticity of the 2 electrons in the π-system; and Baird aromaticity of the 4 electrons in the σ-radial system (SOMO, SOMO’ and HOMO-2).
To further confirm the Baird aromaticity of the triplet Al44-, the researchers calculated the singlet-triplet energy gaps of both the naked anion and the cation stabilised species, Li3Al4–. They noted small energy gaps between the singlet and triplet states, which most likely results from the extra stabilisation of the Baird aromaticity in the triplet state. The researchers also performed an electron density of delocalised bonds (EDDB) analysis to quantify the extent of electron delocalisation (and therefore aromatic character) of Al44-, Li3Al4– and other metallic systems, in comparison to a classical Baird aromatic organic molecule, cyclobutadiene (CBD). They noted comparable values for π-electron delocalisation in the metallic systems compared to CBD, indicating triplet state aromaticity. Additional calculations carried out by the researchers further proved the Baird aromatic character of the all-metal systems, demonstrating how the concept of Baird aromaticity can be extended beyond organic systems and paving the way for future understanding of aromatic, excited state metallic systems.
To find out more, please read:
Dandan Chen, Dariusz W. Szczepanik, Jun Zhu and Miquel Solà
Chem. Commun., 2020, 56, 12522-12525
About the blogger:
 Dr. Samantha Apps just finished her post as a Postdoctoral Research Associate in the Lu Lab at the University of Minnesota, USA, and obtained her PhD in 2019 from Imperial College London, UK. She has spent the last few years, both in her PhD and postdoc, researching synthetic nitrogen fixation and transition metal complexes that can activate and functionalise dinitrogen. Outside of the lab, you’ll likely find her baking at home, where her years of synthetic lab training has sparked a passion in kitchen chemistry too.
Dr. Samantha Apps just finished her post as a Postdoctoral Research Associate in the Lu Lab at the University of Minnesota, USA, and obtained her PhD in 2019 from Imperial College London, UK. She has spent the last few years, both in her PhD and postdoc, researching synthetic nitrogen fixation and transition metal complexes that can activate and functionalise dinitrogen. Outside of the lab, you’ll likely find her baking at home, where her years of synthetic lab training has sparked a passion in kitchen chemistry too.