Solar-driven water reduction is a sustainable method to acquire hydrogen fuel. An indispensable component of this reaction is the photocatalyst which drives spontaneous hydrogen gas evolution from water when illuminated. Hetero-structured materials consisting of two or more catalysts stand out as promising hydrogen evolution catalysts, due to the combined advantages of their constituents (e.g. enhanced light-absorption capability). Unfortunately, the weak adhesion between different components is the Achilles heel of conventional hetero-structured photocatalysts. It impedes electron transport from the photocatalysts to the nearby water molecules, hindering the catalytic activity.
A research group led by Xiao Xiao and Jian-Ping Zou from Nanchang Hangkong University of China has demonstrated a solution to the aforementioned challenge. They firmly connected two photocatalysts – Pt-loaded carbon nitride (CN) and the covalent organic framework CTF-1 – via amide bonds, resulting in a new type of hetero-structured photocatalyst, CN/CTF-1, which exhibited a hydrogen evolution rate approximately 3 times faster than those of conventional hetero-structured photocatalysts made of weakly bound CN and CTF-1.
The researchers adopted a two-step method to synthesize CN/CTF-1. They first reacted CTF-1 sheets with 4-aminobenzoic acid to graft carboxylic groups onto the surfaces of the CTF-1 sheets. A subsequent amide condensation between the amine groups of the CN and the carboxyl groups on the CTF-1 bridged the two components. The amide groups serve as electron transport pathways and facilitate the movement of photo-excited electrons from CTF-1 to CN (Figure 1a) which liberates hydrogen gas.
The covalent amide “bridges” gave CN/CTF-1 a fast hydrogen production rate. Quantitatively, when irradiated with a 300 W Xe lamp at 160 mW/cm2, CN/CTF-1 produced ~4 mmol H2 per gram of CN/CTF-1 after 4 h (0.85 mmol H2 h-1 gcatalyst-1), whereas under identical conditions, weakly adhered CN and CTF-1 sheets as well as a physical mixture of CN and CTF-1 all achieved H2 evolution rates of ~1 mmol H2 per gram of photocatalyst (0.30 mmol H2 h-1 gcatalyst-1) (Figure 1b).
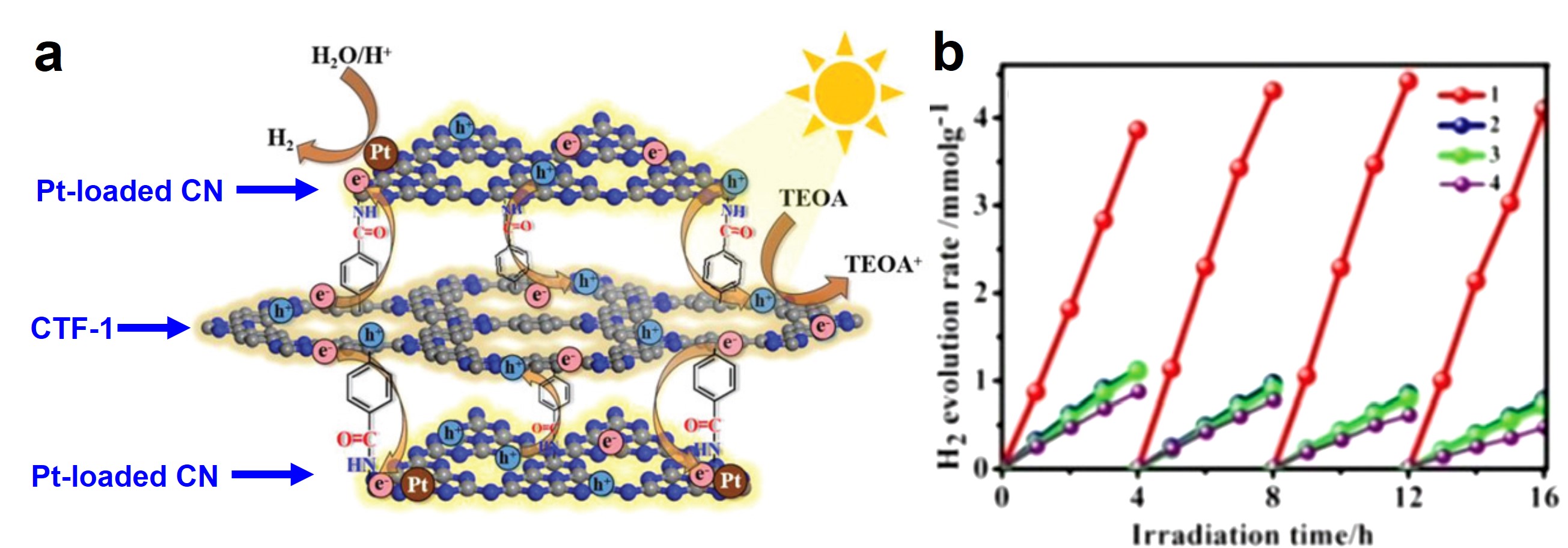
Figure 1. (a) (Pt-loaded) CN sheets are covalently bound to CTF-1 sheets via amide bonds. These covalent bonds serve as electron transport “bridges” that facilitate the diffusion of photo-excited electrons from CTF-1 to CN. (b) H2 evolution rates of four photocatalysts: 1 – covalently bound CN/CTF-1; 2 and 3 – weakly adhered CN and CTF-1; 4 – a physical mixture of CN and CTF-1.
The covalent bonding strategy is applicable to other coupling reactions such as the Friedel-Crafts reaction. This general method could create a new paradigm for designing and synthesizing high-performance hetero-structured photocatalysts.
To find out more please read:
Gang Zhou, Ling-Ling Zheng, Dengke Wang, Qiu-Ju Xing, Fei Li, Peng Ye, Xiao Xiao, Yan Li, and Jian-Ping Zou
Chem. Commun., 2019, 55, 4150-4153
About the blogger:
 Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.
Tianyu Liu obtained his Ph.D. (2017) in Chemistry from University of California, Santa Cruz in the United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is a blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.










