Recently in Chem. Commun., Wang et al. from University of Manchester and Liverpool John Moores University, U.K. demonstrated that in-situ electron paramagnetic resonance (EPR) spectroscopy was a powerful tool to study the charge storage mechanism of activated carbon.
Activated carbon is a type of microporous carbon used for electrodes of supercapacitors (a family of charge-storage devices similar to batteries). Conventional electrochemical testing techniques (e.g. cyclic voltammetry) are able to evaluate the overall performance of electrode materials but are unable to reveal the charge storage mechanism at the atomic level. Understanding the charge storage mechanism is crucial to guide the design and synthesis of electrode materials with improved performance. During the past decade, the development of numerous in-situ probing techniques has allowed materials researchers to explore the microscopic charge-discharge behaviour of supercapacitor electrodes.
In the published paper, in-situ EPR spectroscopy was used to study the electrochemical properties of activated carbon under different external potentials. EPR is very sensitive to electron spins originating from unpaired electrons that are generated upon charging or discharging electrode materials. This characteristic makes EPR a suitable technique for in-situ studies. To carry out the experiments, the authors designed and constructed a capillary three-electrode testing cell (Figure 1a). This cell was placed in an EPR spectrometer and its activated carbon electrode was connected to an external power source (to apply external potentials to the activated carbon electrode). The authors collected the spectra of the activated carbon electrode at selected applied potentials, an example of which is shown in Figure 1b.
Analysis of the obtained spectra offered important information about how the surface of activated carbon changed at different potentials. Specifically, the authors deconvoluted the signal into two components: the narrow signal and the broad signal corresponding to the blue and red curves in Figure 1b, respectively. The peak intensity of the narrow signal increased drastically when charging the electrode, but remained almost unchanged when altering the testing temperature. This observation suggests that the origin of the narrow peaks was the surface-localized electrons. These localized electrons were likely from the oxidized products (i.e. radicals) of carboxylate and alkoxide groups on the surface of the activated carbon, evolved during the charging process. The broad signal was ascribed to electrons located on aromatic units (e.g. graphene domains) and its intensity was found to be proportional to the number of ions electrically adsorbed on the activated carbon surface.
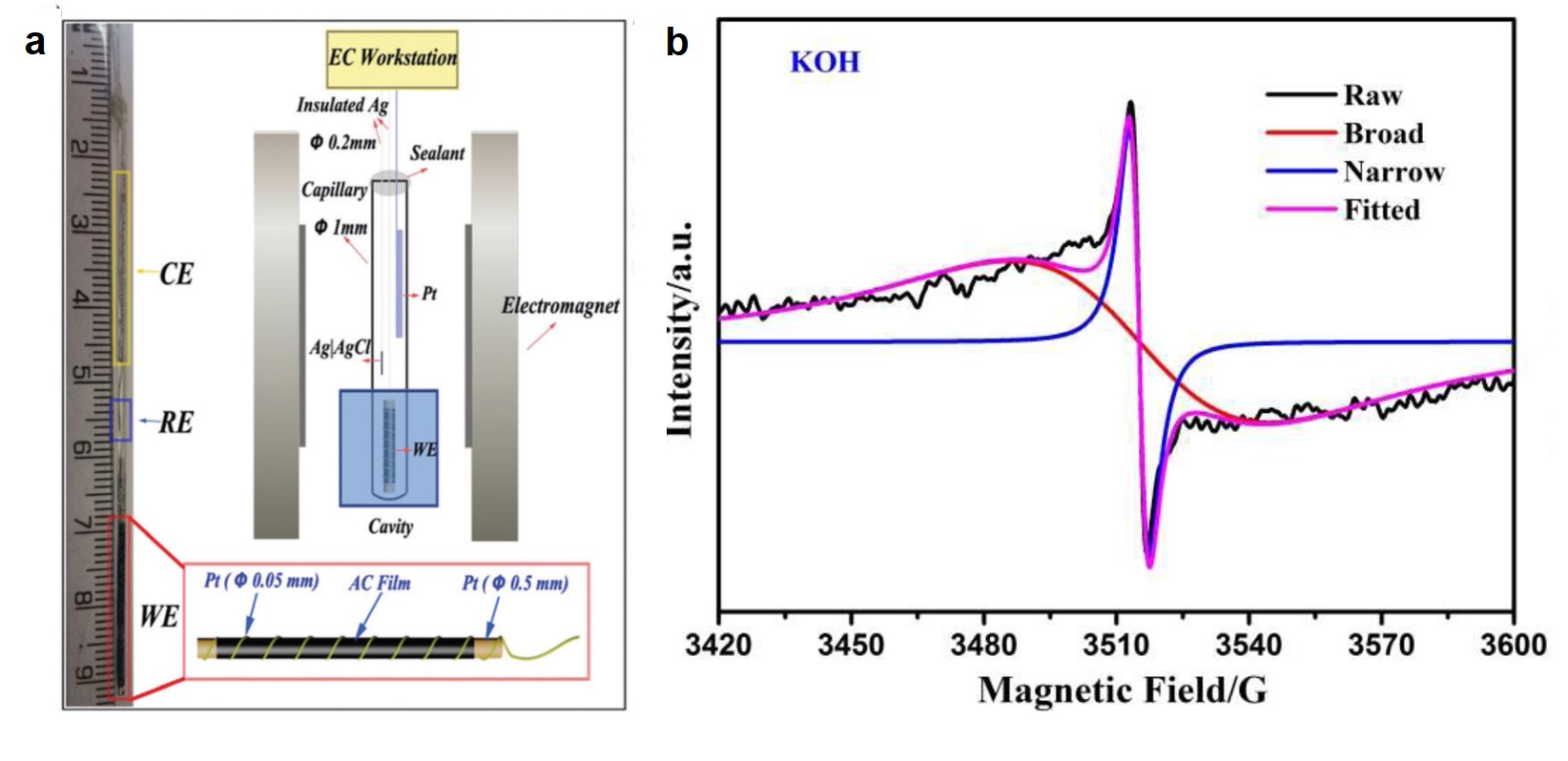
Figure 1. (a) The structure of the self-built capillary three-electrode cell: CE – counter electrode (Pt wire); RE – reference electrode (Ag/AgCl); WE – working electrode (activated carbon). (b) A typical EPR signal (black) that can be deconvoluted into narrow peaks (blue) and broad peaks (red).
This work highlights EPR spectroscopy as a novel tool for in-situ investigation of the charge-storage mechanism of carbon-based supercapacitor electrodes, and could be potentially extended to study other types of materials. The availability of diverse in-situ techniques is expected to provide more in-depth fundamental understanding that will guide researchers to rationally develop electrodes with optimized performance.
To find out more please read:
Bin Wang, Alistair J. Fielding and Robert A. W. Dryfe
Chem. Commun. 2018, DOI: 10.1039/c8cc00450a
About the blogger:
 Tianyu Liu obtained his Ph.D. (2017) in Physical Chemistry from University of California, Santa Cruz in United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is an online blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.
Tianyu Liu obtained his Ph.D. (2017) in Physical Chemistry from University of California, Santa Cruz in United States. He is passionate about scientific communication to introduce cutting-edge research to both the general public and scientists with diverse research expertise. He is an online blog writer for Chem. Commun. and Chem. Sci. More information about him can be found at http://liutianyuresearch.weebly.com/.










