The improvement of high energy density materials (HEDM) is an ongoing challenge. These materials are widely used in propellants, explosives, and pyrotechnics, and researchers face the difficult task of optimising their explosive potential while ensuring their safety and ease of handling. Nitro-substituted methanide compounds are an important class of HEDM, but often suffer from thermal instability and impact sensitivity. This HOT ChemComm article addresses this challenge by highlighting the preparation and analysis of impact insensitive dinitromethanide salts.
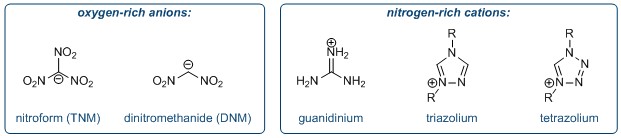
Jean’ne Shreeve at the University of Idaho, working with Ling He at Sichuan University and co-workers at the US Naval Research Laboratory, proposed that by combining an oxygen-rich polynitromethanide anion (either a nitroform anion TNM, or a dinitromethanide anion DNM) with nitrogen-rich cations such as guanidinium, triazolium and tetrazolium anions, the resulting salt would exhibit high energetic properties as well as improved stability.
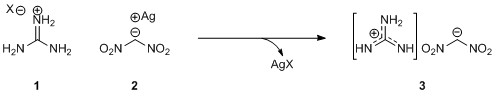
Using a range of guanidinium, triazolium and tetrazolium halides, the researchers prepared nine DNM salts and analysed their physicochemical properties. All of the salts displayed good thermal and detonation properties while being significantly less sensitive to impact than common explosives such as 2,4,6-trinitrotoluene (TNT) and cyclotrimethylenetrinitramine (RDX).
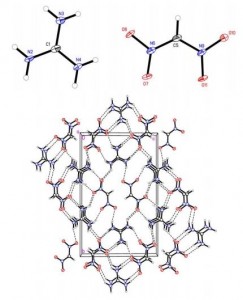
Molecular structure and Packing diagram of DNM salt 3
Guanidinium–DNM salt 3, decomposing at 187 °C, displayed the best thermal stability among all other known DNM salts. X-ray crystallography revealed that this increased stability is due to its strongly hydrogen-bonded structure. Each guanidinium cation forms six hydrogen bonds with the NO2 groups of four surrounding anions, creating a planar, layered packing structure.
Insights such as these will allow researchers to design HEDM with better thermal stability and less impact sensitivity, controlling their energetic potential yet ensuring greater safety and utility.
For more, check out the ChemComm article in full:
Impact insensitive dinitromethanide salts
Ling He, Guo-Hong Tao, Damon A. Parrish, and Jean’ne M. Shreeve
Chem. Commun., 2013, Accepted Manuscript
DOI: 10.1039/C3CC46518G
Ruth E. Gilligan is a guest web-writer for ChemComm. She has recently completed her PhD in the group of Prof. Matthew J. Gaunt at the University of Cambridge, focusing on the development and application of C–H functionalisation methodology.










