This month sees the following articles in ChemComm that are in the top ten most accessed:-
Two metal–organic frameworks based on a double azolium derivative: post-modification and catalytic activity
Guo-Qiang Kong, Xuan Xu, Chao Zou and Chuan-De Wu
Chem. Commun., 2011, 47, 11005-11007, DOI: 10.1039/C1CC14393J
Design and in situ synthesis of a Cu-based porous framework featuring isolated double chain magnetic character
Chandan Dey, Raja Das, Binoy Krishna Saha, Pankaj Poddar and Rahul Banerjee
Chem. Commun., 2011, 47, 11008-11010, DOI: 10.1039/C1CC13418C
Enzyme-free colorimetric assay of serum uric acid
Raj Kumar Bera, Anakuthil Anoop and C. Retna Raj
Chem. Commun., 2011, 47, 11498-11500, DOI: 10.1039/C1CC13349G
Hydrogen bonded supramolecular polymers in moderately polar solvents
Farid Ouhib, Matthieu Raynal, Benjamin Jouvelet, Benjamin Isare and Laurent Bouteiller
Chem. Commun., 2011, 47, 10683-10685, DOI: 10.1039/C1CC14590H
Enantioselective synthesis of 2-methyl indolines by palladium catalysed asymmetric C(sp3)–H activation/cyclisation
Saithalavi Anas, Alex Cordi and Henri B. Kagan
Chem. Commun., 2011, 47, 11483-11485, DOI: 10.1039/C1CC14292E
Highly tunable arylated cinchona alkaloids as bifunctional catalysts
Cormac Quigley, Zaida Rodríguez-Docampo and Stephen J. Connon
Chem. Commun., 2012, Advance Article, DOI: 10.1039/C1CC14684J
A FRET-based indicator for imaging mitochondrial zinc ions
Kesavapillai Sreenath, John R. Allen, Michael W. Davidson and Lei Zhu
Chem. Commun., 2011, 47, 11730-11732, DOI: 10.1039/C1CC14580K
Photo-driven anti-Markovnikov alkyne hydration in self-assembled hollow complexes
Takashi Murase, Hiroki Takezawa and Makoto Fujita
Chem. Commun., 2011, 47, 10960-10962, DOI: 10.1039/C1CC14523A
Nitroolefin-based coumarin as a colorimetric and fluorescent dual probe for biothiols
Yuan-Qiang Sun, Maliang Chen, Jing Liu, Xin Lv, Jun-fei Li and Wei Guo
Chem. Commun., 2011, 47, 11029-11031, DOI: 10.1039/C1CC14299B
Potassium tert-butoxide mediated Heck-type cyclization/isomerization–benzofurans from organocatalytic radical cross-coupling reactions
Magnus Rueping, Matthias Leiendecker, Arindam Das, Thomas Poisson and Lan Bui
Chem. Commun., 2011, 47, 10629-10631, DOI: 10.1039/C1CC14297F
Why not take a look at the articles today and blog your thoughts and comments below.
Fancy submitting an article to ChemComm? Then why not submit to us today or alternatively contact us your suggestions.















 ….and did you know?….
….and did you know?….
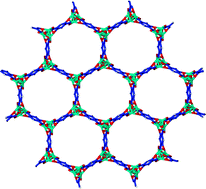 Xenon is naturally present in very small amounts in the atmosphere but radioactive forms are released following nuclear detonations, reprocessing and explosions, such as the recent catastrophe at Fukushima Daiichi Nuclear Power Plant in Japan. Xenon is also used in a variety of other applications, from lighting to medical imaging, so capturing and separating it (from its sister noble gas krypton) is important for both commercial uses and atmospheric monitoring.
Xenon is naturally present in very small amounts in the atmosphere but radioactive forms are released following nuclear detonations, reprocessing and explosions, such as the recent catastrophe at Fukushima Daiichi Nuclear Power Plant in Japan. Xenon is also used in a variety of other applications, from lighting to medical imaging, so capturing and separating it (from its sister noble gas krypton) is important for both commercial uses and atmospheric monitoring.