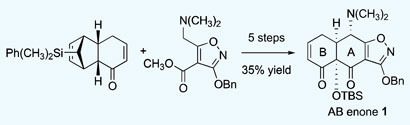
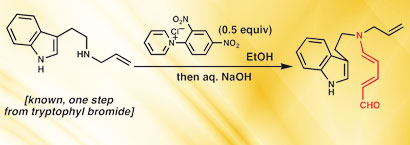
The Royal Society of Chemistry, in collaboration with A*STAR’s Institute of Chemical and Engineering Sciences, GlaxoSmithKline R&D, and with support from the British High Commission in Singapore are organising a symposium on “Contemporary Strategies and Practices in Medicinal Chemistry” to bring together researchers from the United Kingdom and the region to discuss current progress and challenges within the field of medicinal chemistry.
Medicinal chemistry lies at the critical interface between biology and chemistry and plays an integral part of the drug discovery process. The symposium will focus on the current challenges faced by medicinal chemists and feature expert speakers from both the pharmaceutical/biotech industry and leading academic research institutes. The scientific programme will cover some of the very latest chemistry approaches to drug discovery including fragment based hit identification, the application of Click Chemistry, the systematic exploration of chemical space, new approaches to the optimisation of pharmacokinetic and toxicological properties, and progress towards the development of new treatments of cancer and dengue.
In addition to the scientific lectures there will also be a poster session. Abstracts for poster are welcomed and should be submitted to uksin_medchem2011@ices.a-star.edu.sg before 15 August 2011. This free symposium is aimed at researchers in industry and academia, including graduate students and will provide participants with an excellent opportunity to meet and network with potential collaborators.
For registration and more information, please visit http://www.ices.a-star.edu.sg/events/uk-sin_medchem_2011.aspx