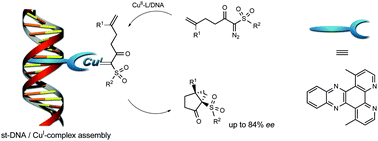
Scientists in the UK are attempting to understand the host–guest chemistry of molecular cages in solution, by separately quantifying the free energy contributions of (at least) three host–guest interaction mechanisms that usually work in unison: hydrogen-bonding, non-polar interactions and solvophobic effects.
The team have systematically looked at a series of guests (with/without π-stacking ability and different hydrogen-bond acceptor capabilities), within a water-soluble cubic coordination cage and an isostructural organic-soluble cage.
Being able to quantify solvent effects associated with specific molecular substituents or functional groups will help in the design of hydrophobic cavities within coordination cages. Hydrophobic cavities can stabilise otherwise unstable molecular guests and catalyse reactions where the transition state matches the cavity size and shape.
Read this ‘HOT’ article, hot off the press:
Quantification of solvent effects on molecular recognition in polyhedral coordination cage hosts
Martina Whitehead , Simon Turega , Andrew Stephenson , Chris Hunter and Mike D Ward
Chem. Sci., 2013, Accepted Manuscript
DOI: 10.1039/C3SC50546D











 Read the ‘HOT’ Chemical Science article for free:
Read the ‘HOT’ Chemical Science article for free: