Source: © Royal Society of Chemistry
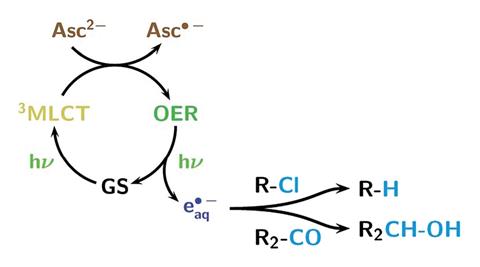
Laboratory-scale photoredox catalysis using hydrated electrons sustainably generated with a single green laser. Asc = ascorbate dianion, OER = one-electron reduced form, GS = ground state, MLCT = metal-to-ligand charge-transfer excited state
Solvated electrons are highly reductive so can force stubborn compounds to react where other reagents might fail. Now a team in Germany is generating them sustainably, using only a green laser and vitamin C. The group has used the technique to perform reactions previously impossible with a visible-light responsive catalyst.
Photons from the laser transform a ruthenium-based photocatalyst into an excited state, with a central oxidised ruthenium atom and a radical anion ligand. Vitamin C’s ascorbate dianion then quenches the excited species, producing a one-electron reduced form of the complex – where the ruthenium atom is no longer oxidised, but the ligand remains as a radical anion. Another photon can then eject the additional electron – generating the hydrated electron species that can induce reduction reactions of organic compounds, and simultaneously returns the catalyst to its ground state.
Read the full story by Jamie Durrani on Chemistry World.










