Scientists from the UK have used a molecular capsule to control the reactivity of an organic compound.
A molecule’s reactivity is commonly controlled using protecting group chemistry, in which a certain functional group is blocked to avoid unwanted reactions at that site. However, this can be costly in terms of time and reagents.
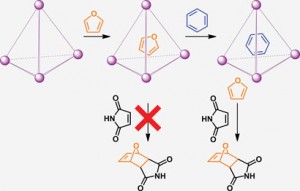
Now, Maarten Smulders and Jonathan Nitschke from the University of Cambridge have used a supramolecular cage as a whole-molecule protecting group. An organic molecule can be encapsulated within the cage, preventing its reaction, and addition of a competing guest can then release the molecule and initiate the reaction when required.
The team constructed the cage from four iron units in a tetrahedral arrangement. External sulfonate groups confer water solubility, while the interior of the cage provides a hydrophobic environment to encapsulate organic molecules. Unlike traditional protecting group chemistry, this method relies not on the functionality of the molecule but on its size and shape, providing a complementary protection strategy to those commonly used.
Encapsulation of furan prevents its reaction, but upon addition of benzene as a competing guest, furan is released from the cage and undergoes a Diels-Alder reaction with maleimide
Read the full story in Chemistry World
Link to journal article
Supramolecular control over Diels–Alder reactivity by encapsulation and competitive displacement
Maarten M. J. Smulders and Jonathan R. Nitschke
Chem. Sci., 2012, Advance Article
DOI: 10.1039/C1SC00847A