Polymer Chemistry issue 9 is now online.
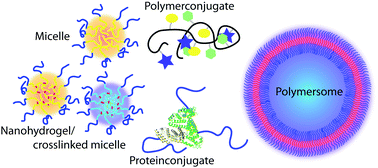
The article highlighted on the front cover is Modular design for the controlled production of polymeric nanotubes from polymer/peptide conjugates by Robert Chapman, Katrina A. Jolliffe and Sébastien Perrier. The paper describes the preparation of nanotubes via the self assembly of well defined peptide polymer conjugates, synthesised using CuAAC chemistry.
Issue 9 contains 4 reviews:
- Overcoming the PEG-addiction: well-defined alternatives to PEG, from structure–property relationships to better defined therapeutics: Matthias Barz, Robert Luxenhofer, Rudolf Zentel and María J. Vicent, Polym. Chem., 2011, 2, 1900-1918
- Fluorene-containing cardo polymers as ion conductive membranes for fuel cells: Kenji Miyatake, Byungchan Bae and Masahiro Watanabe, Polym. Chem., 2011, 2, 1919-1929
- Topological polymer chemistry: a cyclic approach toward novel polymer properties and functions: Takuya Yamamoto and Yasuyuki Tezuka, Polym. Chem., 2011, 2, 1930-1941
- Polymeric chiral organocatalysts: Shinichi Itsuno, Md. Masud Parvez and Naoki Haraguchi, Polym. Chem., 2011, 2, 1942-1949
To keep up-to-date with all the latest research, sign up for the journal’s e-alerts or RSS feeds or follow Polymer Chemistry on Twitter or Facebook.