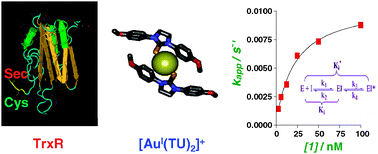
Things are hotting up for ChemComm. The high-pressure, high-temperature explosive RDX (1,3,5-trinitrohexahydro-1,3,5-triazine) is a widely used military explosive that can be compounded with mineral jelly or polymers to form plastic explosives. Colin Pulham from the University of Edinburgh and other collaborators from around the UK have structurally characterised RDX by using a combination of diffraction techniques and successfully recovered a sample at ambient pressure.
This kind of information can be used by scientists to explore aspects of energetic materials (propellants and explosives) that include; sensitivity to shock, heat, and friction; chemical decomposition mechanisms; energy transfer through the solid; detonation velocities; and testing the efficacy of theoretical modelling techniques.
Would you like to read more? Why not read the ChemComm article here, which is free to access until the end of August.