Stimuli-responsive nanoparticles are the focus of much current research, and what could be better than a nanoparticle that responds to one stimulus? A nanoparticle which responds to two or three.
Xianmao Lu and his team have coupled plasmonic silver nanoparticles to magnetic iron oxide nanoparticles and wrapped both in a thermoresponsive polymer – poly(n-isopropylacrylamide).
When illuminated by sunlight the silver nanoparticles absorb the light and convert it to heat. The increase in temperature causes the polymer wrapping to collapse and reduces steric repulsion between the nanoparticle dimers leading to clustering.
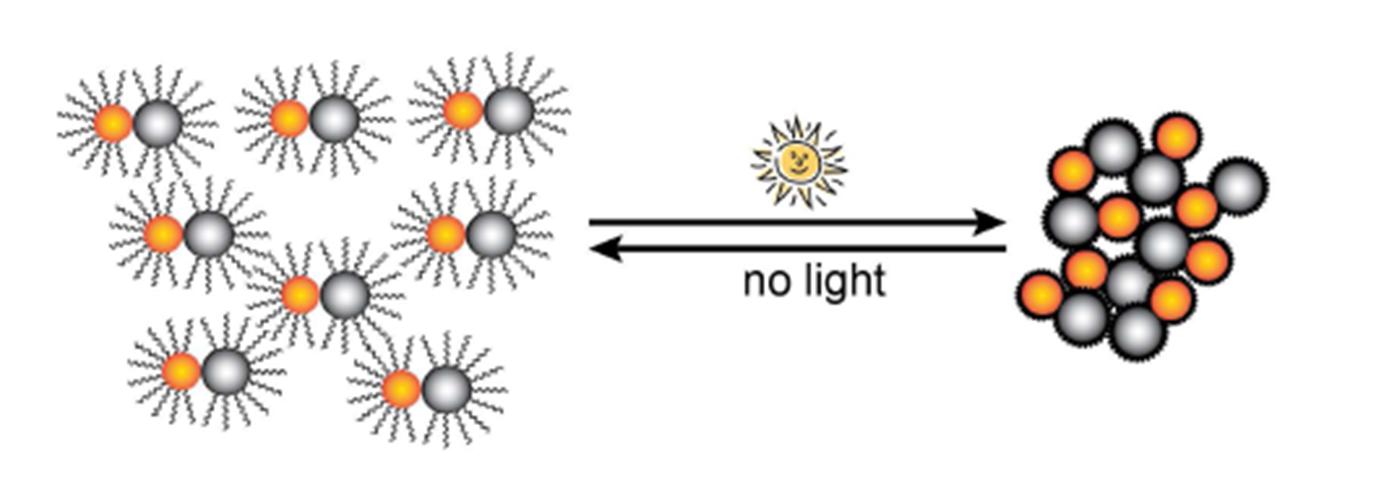
Sunlight induced clustering of Magnetic-Plasmonic Heterodimers.
This clustering enhances the magnetic separation of the very small dimers from the solution (the nanoparticles are less than 9 nm each). When you’ve caught the nanoparticles and are done with them, you can turn the lights off and they will re-disperse.
Don’t worry if you live in a cloudy part of the world, you can use a solar simulator to induce the clustering. It would probably be easier to turn off than the sun, too.
To read the details, check out this HOT Chem Comm article in full:
Thermoresponsive Nanoparticles + Plasmonic Nanoparticles = Photoresponsive Heterodimers: Facile Synthesis and Sunlight-Induced Reversible Clustering
Hui Han, Jim Yang Lee and Xianmao Lu
Chem. Commun., 2013, 49, Accepted Manuscript
DOI: 10.1039/C3CC42273A










