Microorganisms have a remarkable ability to adhere to virtually any type of abiotic surface, proliferate and subsequently form biofilms. Since materials may need to interact differently with microorganisms, the adherent or repellent properties of materials towards bacteria are an extremely important consideration in their design. For example, materials used as implants need to remain free from contamination, in order to reduce device-associated infections, and hence, antifouling surfaces are prepared using water repellent polymers or by anchoring antimicrobial compounds. Conversely, surfaces capable of controlled immobilization and removal of microorganisms have been equally explored for a rather broad range of applications, including sophisticated systems such as biosensors or biomolecular motors. Thus far, precise immobilisation of microorganisms, and in particular bacteria, onto surfaces has been achieved by using a range of different fabrication approaches. However, there are relatively few examples of controlled immobilisation of single bacteria that don’t involve expensive approaches or time-consuming multistep procedures.
In this paper, Palacios-Cuesta and colleagues describe the development of different surface patterns using a photolithographic-based technique that does not require the use of high resolution masks or clean rooms and produces surface patterns with micrometer and submicrometer resolution. The procedure is based on the cross-linking and degradation processes occurring in polystyrene upon exposure to UV light. Together, these processes produce different patterns depending, not only on the mask, but also on the experimental conditions employed. With this approach it is possible to produce patterns with nanoscale resolution without expensive fine focalisation settings. Of particular interest, the authors demonstrate the feasibility of this strategy to incorporate functional groups to modulate the affinity between the bacteria and the surface. In particular, hydrophilic segments, i.e. poly(acrylic acid) that favour bacterial immobilisation are introduced. The strategy employed allows not only the incorporation of functional groups, but also enables the fine tuning of the amount of hydrophilic functional groups. This unique feature is used by the authors to determine the role of surface hydrophilicity on the adhesion of Staphylococcus aureus onto the different surface patterns. Finally, those surfaces on which both photodegradation and photo-cross-linking occur produce thin patterns largely below the micrometer that are then used to prepare arrays of isolated S. aureus bacteria. The formation of bacterial arrays of S. aureus on the single-cell level has been a challenge since they exhibit a large tendency to grow in clusters. This technology is exciting given its potential for enabling the isolation of single bacteria for diagnosis, and the study of bacterial populations at the single cell level.
Patterning of individual Staphylococcus aureus bacteria onto photogenerated polymeric surface structures by Marta Palacios-Cuesta, Aitziber L. Cortajarena, Olga García and Juan Rodríguez-Hernández, Polym. Chem., 2015, 6, 2677-2684.
Remzi Becer is a web-writer and Advisory Board member for Polymer Chemistry. He is currently a Senior Lecturer in Materials Science and the director of the Polymer Science and Nanotechnology masters programme at Queen Mary, University of London. Visit www.becergroup.com for more information.












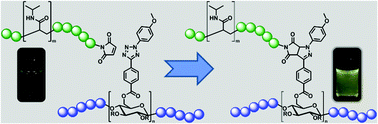
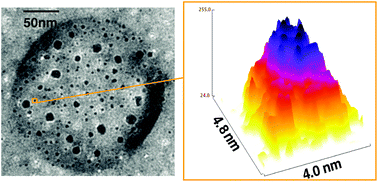
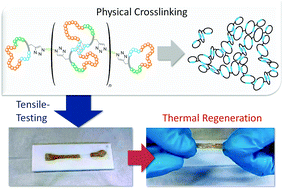
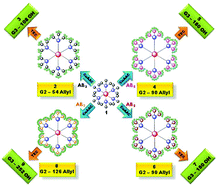
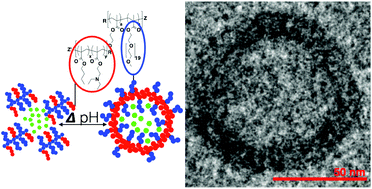
![[similar]](http://www.rsc.org/images/entities/char_223c.gif) 950 Da) (O950) with variable comonomer compositions and molecular weights for use as polymeric scaffolds. Reactivity ratios for the monomer pair were determined to be 1.37 and 0.290 respectively. To these scaffolds, trithiocarbonate-based RAFT chain transfer agents (CTAs) were grafted using carbodiimide chemistry. The resultant graft chain transfer agents (gCTA) were subsequently employed to polymerise dimethylaminoethyl methacrylate (DMAEMA) and N-(2-hydroxypropyl)methacrylamide (HPMA) between degrees of polymerisation (DP) of 25 and 200. Kinetic analysis for the polymerisation of DMAEMA targeting a DP of 100 from a 34 arm graft gCTA show linear Mn conversion and pseudo first order rate plots with narrow molecular weight distributions that move toward lower elution volumes with monomer conversion. Đ values for these polymerisations remain low at around 1.20 at monomer conversions as high as 70%. pH-responsive endosomalytic brushes capable of spontaneously self-assembling into polymersomes were synthesised and a combination of dynamic light scattering (DLS), cryoTEM, and red blood cell haemolysis were employed to evaluate the aqueous solution properties of the polymeric brush as a function of pH. Successful encapsulation of ceftazidime and pH-dependent drug release properties were confirmed by HPLC. Intracellular antibiotic activity of the drug-loaded polymersomes was confirmed in a macrophage coculture model of infection with Burkholderia thailandensis and RAW 264.7 cells.
950 Da) (O950) with variable comonomer compositions and molecular weights for use as polymeric scaffolds. Reactivity ratios for the monomer pair were determined to be 1.37 and 0.290 respectively. To these scaffolds, trithiocarbonate-based RAFT chain transfer agents (CTAs) were grafted using carbodiimide chemistry. The resultant graft chain transfer agents (gCTA) were subsequently employed to polymerise dimethylaminoethyl methacrylate (DMAEMA) and N-(2-hydroxypropyl)methacrylamide (HPMA) between degrees of polymerisation (DP) of 25 and 200. Kinetic analysis for the polymerisation of DMAEMA targeting a DP of 100 from a 34 arm graft gCTA show linear Mn conversion and pseudo first order rate plots with narrow molecular weight distributions that move toward lower elution volumes with monomer conversion. Đ values for these polymerisations remain low at around 1.20 at monomer conversions as high as 70%. pH-responsive endosomalytic brushes capable of spontaneously self-assembling into polymersomes were synthesised and a combination of dynamic light scattering (DLS), cryoTEM, and red blood cell haemolysis were employed to evaluate the aqueous solution properties of the polymeric brush as a function of pH. Successful encapsulation of ceftazidime and pH-dependent drug release properties were confirmed by HPLC. Intracellular antibiotic activity of the drug-loaded polymersomes was confirmed in a macrophage coculture model of infection with Burkholderia thailandensis and RAW 264.7 cells.