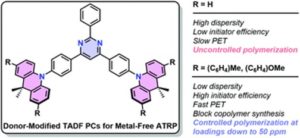
Polgar et al. use donor-modified thermally activated delayed fluorescence (TADF) emitters as photocatalysts for O-ATRP.
O-ATRP has emerged as an attractive alternative to conventional metal-catalyzed ATRP that uses organic compounds that catalyze polymerization under light irradiation instead of the well-studied metal catalysts. Specifically, O-ATRP using thermally activated delayed fluorescence (TADF) emitters as photocatalysts takes advantage of the unique ability of TADFs to interconvert singlet and triplet excited states and has been more recently implemented in diverse fields including organic electronics, photocatalysis, biological imaging, and chemical sensing. TADF emitters mostly contain a twisted donor–π–acceptor (D–π–A) motif which results in a prolonged excited state lifetime and facilitates singlet and triplet energy and electron transfer. Nevertheless, the coexistence of both electron donors and acceptors in photoredox catalysis results in unwanted excited state side reactivity that limits initiator efficiency and might also deactivate the catalyst.
To address this issue, Hudson and collaborators use donor-modified TADF emitters as photocatalysts for O-ATRP. More specifically, TADF photosensitizers based on 9,10-dihydro-9,9-dimethylacridine/2,4,6-triphenylpyrimidine conjugates exhibiting strong visible absorption, large excited state reduction potentials, and long-lived triplet excited states were employed to evaluate catalyst structure–activity relationships. The stability of the radical cation was found to be determining for controlled polymerization, however, significant differences were observed among donor-modified catalysts which were also related to variation in the rates of photoinduced electron transfer (PET). Time-resolved photoluminescence studies of the catalysts supported initiation by electron transfer from both singlet and triplet states while, the functionalized donors possessed the higher driving forces for PET. Through this study, the donor-modified TADF photocatalyst PymDMDMA -bearing a methoxyphenyl substituent- was identified to yield methacrylic polymers with Đ below 1.3 at low catalyst loadings (50 ppm) while also being able to catalyze the controlled synthesis of block copolymers in contrast to the unmodified TADF.
This study explores the ability to design more efficient catalysts by merely altering the types of donors, acceptors and their derivatives and proposes that more efficient catalysts can be designed through theoretical modeling.
Tips/comments directly from the authors:
- ppm-levels of catalyst are sufficient for the synthesis of polymers with well-defined size, composition, and topology by O-ATRP, potentially obviating the need for post-polymerization purification.
- Donor-acceptor fluorophores can exhibit thermally activated delayed fluorescence (TADF), a phenomenon that prolongs the excited state lifetime. The ability to control the lifetime and excited-state reduction potential of TADF emitters makes them versatile photocatalysts for O-ATRP, particularly at low catalyst loadings.
- A donor-modification strategy was used in this study to mitigate deleterious excited-state side-reactivity associated with the electron-rich donors used in TADF. Rational design of modifying groups can not only enhance the photostability of these dyes, but also provide an extra dimension of control over the excited state reduction potentials and rates of electron transfer in O-ATRP.
Donor modification of thermally activated delayed fluorescence photosensitizers for organocatalyzed atom transfer radical polymerization, Polym. Chem., 2022, 13, 3892-3903.
Link to the paper: https://pubs.rsc.org/en/content/articlelanding/2022/py/d2py00470d