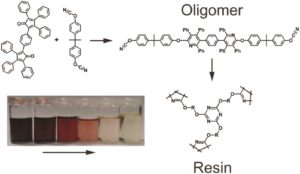
Hyperbranched polyphenylenes and cyanate esters are two unique classes of materials that possess complementary properties. On one side, polyphenylenes are good insulators with remarkable solubility owing to their dense packing and the strongly twisted structure hinder π-conjugation respectively. Cyanate esters are also well renowned for their thermal stability as thermosetting materials. To combine these properties, Voit and co-workers investigated the copolymerisation of the two monomers 3,3′-(1,4-phenylene)bis(2,4,5-triphenylcyclo-pentadienone) and 2,2-bis(4-cyanatophenyl) propane through a Diels-Alder cycloaddition where carbon monoxide is released as a side product. The polymerisation was followed by UV/Vis spectroscopy and the structure of the oligomers could be further investigated by in-depth NMR studies. Importantly, the catenation proved to be completely statistical and independent of the temperature of the polymerization while the obtained oligomers can be cured via a trimerisation reaction of the terminal OCN-groups. Finally, the polymerisation and crosslinking reaction kinetics were also studied and upon crosslinking the resins exhibit high thermal resistance and transparency as well as a high refractive index. Thus, the resulting materials simultaneously possess the strengths of polyphenylene polymers while retaining the curing potential of the cyanate esters but at only the tenth of the activation energy of pure cyanate monomers, lowering the risk factors during handling. As the authors elegantly conclude, materials with such unique characteristics may find application in integrated optics.
Tips/comments directly from the authors:
- The Diels-Alder cycloaddition with these substrates requires high temperatures. However, under these conditions the trimerisation reaction of cyanate esters also takes place. To avoid the premature crosslinking of the system while maintaining the cyanate ester termination a special protocol was developed. During the Diels-Alder reaction the ratios where adjusted to obtain a oligomer terminated with cyclopentadienone groups and only 15 minutes prior to the end of the reaction one additional equiv. of cyanate ester was added.
- The cyclopentadienone possesses a deep purple color while the polymer is colorless. Therefore, UV/Vis spectroscopy can be a powerful tool to track the reaction, but a simple look inside the reaction vial already gives indications on the state of the reaction.
- While cyclopentadienone monomers are sometimes challenging to synthesize there is a wide variety of commercial cyanate ester monomers and prepolymers allowing for a high degree of tunability of the resulting resin without changing the cyclopentadienone unit.
- Different to fully phenylene-based systems which are difficult to analyze by 13C NMR spectroscopy, the reaction with cyanate results in pyridine and cyanurate structures that can be well identified thus improving the structural characterization of such oligomers.
Read the full paper for FREE until 1st April 2019!
A Diels–Alder reaction between cyanates and cyclopentadienone-derivatives – a new class of crosslinkable oligomers, Polym. Chem., 2019, 10, 698-704
About the Web Writer
 Dr. Athina Anastasaki is an Editorial Board Member and a Web Writer for Polymer Chemistry. Since January 2019, she joined the Materials Department of ETH Zurich as an Assistant Professor to establish her independent research group.
Dr. Athina Anastasaki is an Editorial Board Member and a Web Writer for Polymer Chemistry. Since January 2019, she joined the Materials Department of ETH Zurich as an Assistant Professor to establish her independent research group.