With increasing demands upon our planet’s resources, more and more research efforts are focussing on preparing new materials from renewable resources. Especially considering the global plastic production (299 million tonnes in 2013), new routes towards renewable polymers are ever more desirable. Renewable polymers can generally be classified into the following three groups: naturally occurring polymers such as polysaccharides and proteins, polymers prepared by microbial fermentation and polymers synthesised from bioderived monomers. This month, focussing on polymers from renewable resources, we take look at three articles which utilize renewable plant-derived monomers to synthesise various polyesters. The plant-derived monomers described are based on the following compounds: erucic acid, commonly found in rapeseed oil, eugenol which is present in clove oil and δ-decalactone which can be extracted from Cryptocarya massoia.
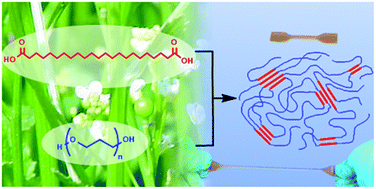
1. Thermoplastic polyester elastomers based on long-chain crystallizable aliphatic hard segments, Florian Stempfle, Brigitta Schemmer, Anna-Lena Oechsle, Stefan Mecking, Polym. Chem., 2015, 6, 7133-7137.
Thermoplastic polyesters were prepared via polycondensation with a plant-oil based long chain (C23) α,ω-dicarboxylic acid and the corresponding diol comprising the hard segments, and poly(tetramethylene glycol) (PTMG) or carbohydrate-based poly(trimethylene glycol) (PPDO) soft segments. Physical crosslinking was achieved through crystallization of the long aliphatic segments, resulting in enhanced thermal properties when compared to the C12 analogues.
2. Synthesis and properties of polyesters derived from renewable eugenol and α,ω-diols via a continuous overheating method, Keling Hu, Dongping Zhao, Guolin Wu, Jianbiao Ma, Polym. Chem., 2015, 6, 7138-7148.
Aromatic monomers derived from eugenol were synthesised utilizing “click” chemistry and a subsequent Williamson ether synthesis. They were subsequently polymerised with various α,ω-diols via a continuous overheating method across the transesterification stage to form polyester thermoplastics. The Young’s modulus and ultimate strength of the resulting materials was not particularly high although they exhibited excellent ductility.
3. New biomaterials from renewable resources – amphiphilic block copolymers from δ-decalactone, Kuldeep K. Bansal, Deepak Kakde, Laura Purdie, Derek J. Irvine, Steven M. Howdle, Giuseppe Mantovani, Cameron Alexander, Polym. Chem., 2015, 6, 7196-7210.
Various polymers and copolymers were synthesized from the low-cost and easily-accessible renewable monomer δ-decalactone, which is an FDA approved flavouring agent. Amphiphilic copolymers were shown to self-assemble into micelles which were biodegradable and showed low toxicity in vitro. The micelles enabled sustained release of a model drug compound over 8 days, highlighting the possible biomedical application of these degradable δ-decalactone copolymers.
Dr. Fiona Hatton is a Web Writer for Polymer Chemistry. She is currently a postdoctoral researcher at KTH Royal Institute of Technology, Sweden, having completed her PhD in the Rannard group at the University of Liverpool, UK. Visit her webpage for more information.