N ate studied chemistry at Michigan State University, and attended graduate school at the University of Minnesota where he worked in the laboratory of Prof. Marc A. Hillmyer on the effects of polydispersity on block polymer self-assembly. After graduating in late 2007, Nate moved to the Materials Research Laboratory at the University of California in Santa Barbara and worked in the groups of Glenn H. Fredrickson, Edward J. Kramer, and Craig J. Hawker. During nearly six years of research at the MRL, Nate published work in areas of polymer science encompassing a broad range of synthetic, physical, and theoretical topics. After UCSB, Nate became a staff scientist at Lawrence Berkeley National Laboratory in the Materials Sciences Division and became a project leader at the Joint Center for Artificial Photosynthesis. In April 2014, Nate was appointed as an assistant professor in the McKetta Department of Chemical Engineering at the University of Texas at Austin.
ate studied chemistry at Michigan State University, and attended graduate school at the University of Minnesota where he worked in the laboratory of Prof. Marc A. Hillmyer on the effects of polydispersity on block polymer self-assembly. After graduating in late 2007, Nate moved to the Materials Research Laboratory at the University of California in Santa Barbara and worked in the groups of Glenn H. Fredrickson, Edward J. Kramer, and Craig J. Hawker. During nearly six years of research at the MRL, Nate published work in areas of polymer science encompassing a broad range of synthetic, physical, and theoretical topics. After UCSB, Nate became a staff scientist at Lawrence Berkeley National Laboratory in the Materials Sciences Division and became a project leader at the Joint Center for Artificial Photosynthesis. In April 2014, Nate was appointed as an assistant professor in the McKetta Department of Chemical Engineering at the University of Texas at Austin.
Nate’s research efforts are focused on creating and utilizing new functional and reactive macromolecular materials. Newer work is built on a foundation of techniques for advanced copolymer structure determination and detailed mechanistic understanding which facilitate the compositional control of structure-property-processing relationships. Specifically, we are currently engaged in gaining further understanding of compositional control in polyether materials, degradable biomedical materials, membranes for carbon capture, and understanding ion transport in polymer electrolytes and membranes.
Research group: http://lynd.che.utexas.edu/
What was your inspiration in becoming a chemist?
I always had an interest in science as a kid. I was (and still am!) very interested in geology, paleontology, and space exploration like many. My interest narrowed to chemistry with a very inspirational honors chemistry teacher in high school (Mr. John Wheeler, Batavia High School). In undergraduate, I took organic chemistry from Dr. Gregory L. Baker, a polymer chemist. I was fascinated by the possibilities of polymers, and did undergraduate research in Dr. Baker’s lab on the stereospecific synthesis of substituted lactides. This was how I got started in polymer science.
What was the motivation to write your Polymer Chemistry article?
Functional, mono-sized polymer particles are enabling to a range of applications. Our interest in this was motivated by a one very specific application, but we focused on the underlying chemistry for the article.
Why did you choose Polymer Chemistry to publish your work?
I’ve had a very positive experience with the journal, and I think it’s the right venue for rapidly reporting new developments. Additionally, I believe it reaches the right audience. As such, I’ve selected Polymer Chemistry to publish several other articles as well.
In which upcoming conferences may our readers meet you?
I will be a several upcoming conferences. I’ll be at an ECI conference on membranes in Syracuse, Italy in February, also the upcoming American Chemical Society Meeting in Denver. Additionally, I’ll be at the upcoming Gordon Research Conference on Polymers in June, and will attend the International Symposium on Ionic Polymerization in Bordeaux this July.
How do you spend your spare time?
I enjoy reading science fiction, spending time with my family, and running whenever I get a chance. I enjoy cooking and especially barbecue. Lately, I’ve been working on my central Texas style BBQ’ing skills!
Which profession would you choose if you were not a scientist?
I would probably enjoy being a scientific illustrator, or a software engineer.
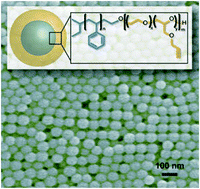
A synthetic strategy for the preparation of sub-100 nm functional polymer particles of uniform diameter
Kato L. Killops, Christina G. Rodriguez, Pontus Lundberg, Craig J. Hawk and Nathaniel A. Lynd
Abstract: An amphiphilic block copolymer surfactant is used to impart peripheral surface functionality to polymer nanoparticles synthesized via emulsion polymerization. Particles ranged in size from ca. 55 nm by SEM (ca. 82 nm by DLS) to just over 200 nm. Particles displaying latent functionality were readily functionalized directly after polymerization using a fluorescent dye.
Cyrille Boyer is a guest web-writer for Polymer Chemistry. He is currently an associate professor and an ARC-Future Fellow in the School of Chemical Engineering, University of New South Wales (Australia) and deputy director of the Australian Centre for NanoMedicine.