Dr. Antoine Debuigne is currently a Research Associate of the National Fund for Scientific Research (F.R.S. – FNRS) at the University of Liege, Belgium. He graduated in 1999 from the University of Namur, Belgium, following a master’s thesis in organic chemistry. In 2004, he obtained his Ph.D. degree from the Center for Education and Research on Macromolecules (CERM), University of Liege, Belgium, directed by Prof. R. Jérôme. He then moved to the University of Toronto, Canada, and conducted post-doctoral research in Prof. M.K. Georges’ group, who pioneered the field of controlled radical polymerisation. In 2006, he returned to the CERM in Liege as a FNRS post-doctoral researcher and was promoted to FNRS Research Associate in 2010 in the same group. His research interests include controlled radical polymerisation, organometallic chemistry, macromolecular engineering and emulsion polymerisation.
Dr. Antoine Debuigne is currently a Research Associate of the National Fund for Scientific Research (F.R.S. – FNRS) at the University of Liege, Belgium. He graduated in 1999 from the University of Namur, Belgium, following a master’s thesis in organic chemistry. In 2004, he obtained his Ph.D. degree from the Center for Education and Research on Macromolecules (CERM), University of Liege, Belgium, directed by Prof. R. Jérôme. He then moved to the University of Toronto, Canada, and conducted post-doctoral research in Prof. M.K. Georges’ group, who pioneered the field of controlled radical polymerisation. In 2006, he returned to the CERM in Liege as a FNRS post-doctoral researcher and was promoted to FNRS Research Associate in 2010 in the same group. His research interests include controlled radical polymerisation, organometallic chemistry, macromolecular engineering and emulsion polymerisation.
To find out more about the CERM please follow the link.
What was your inspiration in becoming a chemist?
Several members of my family are scientists, even chemists, so “I fell early into chemistry”. During my scholarship, I also met excellent science teachers who taught me how important science is for understanding the world we are living in. So, I decided to make it my job. Among all scientific fields, I chose chemistry because it appeared to me as a central and practical discipline.
What was the motivation to write your Polymer Chemistry article?
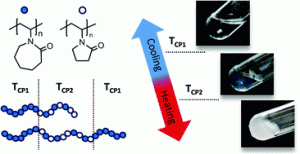
Recent progress in controlled radical polymerisation (CRP) is impressive. The increasing precision and complexity of polymer structures achievable and their use in a broad range of applications might suggest that challenges in CRP may soon be non-existent. However, this is not the case and efforts still need to be expended to improve the existing tools and discover new ones in order to finely tune polymer properties. This is what the present Polymer Chemistry article is about. Indeed, poly(N-vinylcaprolactam) (PNVCL) combines valued properties such as water solubility, biocompatibility and thermo-responsiveness, but has not reached its full potential so far due to limitations in macromolecular engineering techniques. Nevertheless, the Organometallic-Mediated Radical Polymerisation (OMRP), a CRP method developed in the lab for many years, permitted us to produce a series of well-defined N-vinylcaprolactam and N-vinylpyrrolidone-based copolymers including statistical, diblock and triblock copolymers, having single or dual thermo-responsive behaviour in water. The high level of control afforded by OMRP allowed us to highlight the crucial impact of the copolymer composition, block length and architecture on the cloud point temperature of each segment and to tune their multistep assembly behaviour. The reversible temperature triggered assembly of such block copolymers in water opens new perspectives in the field of stimuli-responsive encapsulation/release applications.
Why did you choose Polymer Chemistry to publish your work?
My co-authors and I chose Polymer Chemistry because it provides high quality manuscripts in polymer science to a broad audience. This last aspect was crucial in the selection of a journal because our contribution contains progress in both macromolecular engineering and characterisation of the thermal response of copolymers in solution.
In which upcoming conferences may our readers meet you?
My agenda is still unclear for the next few months but I really would like to attend the Gordon Research Conference on Polymers, South Hadley, MA, USA in June 2015.
How do you spend your spare time?
I enjoy spending time with my 6-year old son, friends and relatives. I also like reading novels and practice more and more photography. In this respect, I warmly recommend a visit to the photography museum of Charleroi in Belgium.
Which profession would you choose if you were not a scientist?
Maybe architect because this profession requires both technical and artistic skills. Having said that, I have no regret at all.
One of my pictures. “Building reflected on water”
Double thermo-responsive hydrogels from poly(vinylcaprolactam) containing diblock and triblock copolymers
Jean-Michel Thomassin, Kevin Mathieu, Anthony Kermagoret, Charles-André Fustin, Christine Jérôme and Antoine Debuigne
The thermally-induced gelation and gel properties of concentrated aqueous solutions of double thermoresponsive poly(N-vinylamide)-based di- and triblock copolymers are studied by rheology. The copolymers under investigation, prepared by cobalt-mediated radical polymerisation and coupling reactions, are composed of poly(vinylcaprolactam) (PNVCL) blocks and of a statistical poly(vinylcaprolactam-stat-vinylpyrrolidone) segment with a cloud point temperature (TCP) higher than that of PNVCL. Heating the di- and triblock solutions beyond the first phase transition temperature favours gel formation while heating above the second TCP leads to opaque gels without macroscopic demixing. Moduli of the triblock hydrogels are systematically higher than those of the corresponding diblocks, even above the second transition. Rheological data suggest distinct micellar structures for each copolymer architecture: densely packed micelles of diblocks and 3-D networks of bridged micelles for triblocks. Strain sweep experiments also emphasize the positive effect of the micelle bridging on the elasticity and stability of the hydrogels. The formation and properties of the obtained gels are also shown to depend on the copolymer concentration, block length, and composition. Addition of salt also allows us to tune the phase transition temperatures of these double thermoresponsive hydrogels.
Cyrille Boyer is a guest web-writer for Polymer Chemistry. He is currently an Associate Professor and an ARC-Future Fellow in the School of Chemical Engineering, University of New South Wales (Australia).