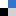 When a droplet impacts on a solid surface it deforms. Depending on the properties of the drop and the surface (velocity, viscosity, surface tension, hydrophobicity etc.) this deformation can be temporary, the droplet spreads out before retracting and bouncing back, or permanent, the drop breaks apart on impact making a splash.
When a droplet impacts on a solid surface it deforms. Depending on the properties of the drop and the surface (velocity, viscosity, surface tension, hydrophobicity etc.) this deformation can be temporary, the droplet spreads out before retracting and bouncing back, or permanent, the drop breaks apart on impact making a splash.
Air also plays an important role in determining the behaviour of the impacting drop. Detlef Lohse, University of Twente, is interested in understanding how and why the air layer profile under a drop influences its deformation as it falls and subsequently hits a surface. His group has developed an ultra-high speed colour interferometry imaging method, allowing them to resolve the dynamics of an impacting drop on short timescales. Using this method, Lohse has been able to measure the time evolution of the drop before and during impact on a solid surface. As the drop falls the air between the drop and surface is strongly squeezed. A region of high pressure builds up, which leads to the formation of a dimple on the underside of the drop. At high velocities this can result in splashing as the air is compressed on impact. The presence of an air bubble can also stop the droplet from touching the surface at all leading to some interesting effects.
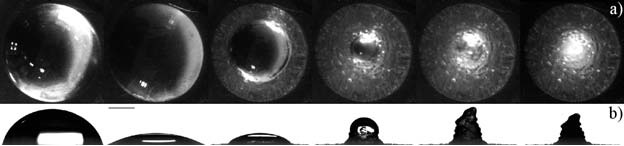
When a liquid droplet impacts a surface heated above the liquid’s boiling point three impact behaviour regimes are observed. In the first, ‘contact boiling’, the droplet immediately boils as it comes into contact with the surface. The droplet contacts the surface and spreads out. Bubbles then form and the liquid evaporates. For ‘gentle film boiling’ the droplet appears to hit the surface before bouncing back. In this regime a Leidenfrost vapour layer forms under the drop before it hits, preventing it from coming into direct contact with the surface. The final behavioural regime is ‘spray film boiling’. In this case, although the droplet does not contact the surface, breakup does occur. A Leidenfrost vapour layer forms below the drop. As the vapour tries to escape it drags fluid out with it and the droplet forms a thinner pancake shape. Tiny drops are the ejected upwards in what can be quite a violent spray event. The impact conditions under which each regime can be observed were recently published in Physical Review Letters.
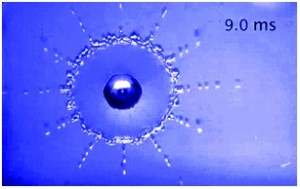
Lohse has also looked at how the structure of the surface can influence the splashing dynamics for high velocity impacting drops. In this case the surfaces are all at room temperature. This is discussed in detail for Newtonian and non-Newtonian liquids in two recent Soft Matter papers. Directional splashing can be tuned and suppressed by varying the periodicity of the lattice and, or the air pressure. A number of videos of the impacting drops can be found in the supplementary information accompanying the Soft Matter article and are well worth a look.