This month sees the following articles in Organic & Biomolecular Chemistry that are in the top ten most accessed:-
Recent advances in transition metal-catalyzed N-atom transfer reactions of azides
Tom G. Driver
Org. Biomol. Chem., 2010, 8, 3831 – 3846, DOI: 10.1039/c005219c, Perspective
Sequential combination of Michael and acetalization reactions: direct catalytic asymmetric synthesis of functionalized 4-nitromethyl-chromans as drug intermediates
Dhevalapally B. Ramachary and Rajasekar Sakthidevi
Org. Biomol. Chem., 2010, 8, 4259 – 4265, DOI: 10.1039/c0ob00189a, Communication
Strain-promoted double-click reaction for chemical modification of azido-biomolecules
Isao Kii, Akira Shiraishi, Toshiyuki Hiramatsu, Takeshi Matsushita, Hidehiro Uekusa, Suguru Yoshida, Makoto Yamamoto, Akira Kudo, Masatoshi Hagiwara and Takamitsu Hosoya
Org. Biomol. Chem., 2010, 8, 4051 – 4055, DOI: 10.1039/c0ob00003e, Communication
Recent advances in stereoselective glycosylation through intramolecular aglycon delivery
Akihiro Ishiwata, Yong Joo Lee and Yukishige Ito
Org. Biomol. Chem., 2010, 8, 3596 – 3608, DOI: 10.1039/c004281a, Perspective
A facile method for the synthesis of oxindole based quaternary a-aminonitriles via the Strecker reaction
Yun-Lin Liu, Feng Zhou, Jun-Jie Cao, Cong-Bin Ji, Miao Ding and Jian Zhou
Org. Biomol. Chem., 2010, 8, 3847 – 3850, DOI: 10.1039/c0ob00174k, Communication
High-throughput synthesis of azide libraries suitable for direct “click” chemistry and in situ screening
Rajavel Srinivasan, Lay Pheng Tan, Hao Wu, Peng-Yu Yang, Karunakaran A. Kalesh and Shao Q. Yao
Org. Biomol. Chem., 2009, 7, 1821 – 1828, DOI: 10.1039/b902338k
In water, on water, and by water: mimicking nature’s aldolases with organocatalysis and water
Nobuyuki Mase, Carlos F. Barbas III
Org. Biomol. Chem., 2010, 8, 4043 – 4050, DOI: 10.1039/c004970k, Perspective
Organosilicon-mediated total synthesis of the triquinane sesquiterpenes (±)-beta-isocomene and (±)-isocomene
Arndt W. Schmidt, Thomas Olpp, Elke Baum, Tina Stiffel and Hans-Joachim Knölker
Org. Biomol. Chem., 2010, DOI: 10.1039/c0ob00051e
Intramolecular cycloaddition in 6,6-spiroepoxycyclohexa-2,4-dienone: simple aromatics to (±)-Platencin
Vishwakarma Singh, Bharat Chandra Sahu, Varsha Bansal and Shaikh M. Mobin
Org. Biomol. Chem., 2010, 8, 4472 – 4481, DOI: 10.1039/c004316h
Radical stability and its role in synthesis and catalysis
Johnny Hioe and Hendrik Zipse
Org. Biomol. Chem., 2010, 8, 3609 – 3617, DOI: 10.1039/c004166a, Perspective
Why not take a look at the articles today and blog your thoughts and comments below.
Fancy submitting an article to Organic & Biomolecular Chemistry? Then why not submit to us today or alternatively email us your suggestions.
Comments Off on Top ten most accessed articles in August


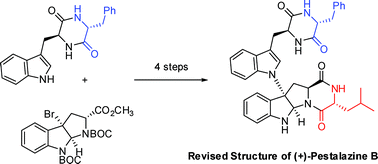









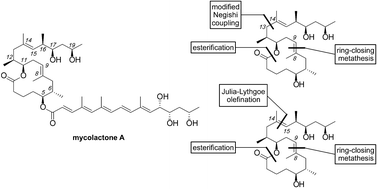 Synthetic studies on the mycolactone core
Synthetic studies on the mycolactone core 
 Hans-Joachim Knölkner and colleagues reveal the link between this beautiful church in Dresden and β-isocomene: silicon
Hans-Joachim Knölkner and colleagues reveal the link between this beautiful church in Dresden and β-isocomene: silicon This colourful and arty cover by Chunling Fu, Shengming Ma and colleagues shows the mechanism of a highly regio- and stereo- selective hydration of 1,2-allenylic sulfoxides.
This colourful and arty cover by Chunling Fu, Shengming Ma and colleagues shows the mechanism of a highly regio- and stereo- selective hydration of 1,2-allenylic sulfoxides.