Nominations are now invited for the 2011 Organic & Biomolecular Chemistry Lecture Award, to be given at the 16th IUPAC International Symposium on Organometallic Chemistry Directed Towards Organic Synthesis (OMCOS 16), held from 24-28 July 2011 in Shanghai, China.
Anyone can make a nomination for the award. All you need to do is send the name of your nominee and a brief justification to the Editor, Dr Richard Kelly. All nominations should take account of the award criteria (below) and the topic of the conference and should be received by Monday 6th December 2010.
This annual award is open to scientists who have made a significant contribution within the scope of OBC and ideally will have an independent academic career of between 8 and 15 years.
The winner, who must be available to give the sponsored lecture, will also be invited to submit a related article for publication in OBC (which will be subject to the usual peer review process). The award, open to anyone fitting these criteria, will cover various expenses incurred at the meeting.
The winner will be selected from the nominees by a panel of judges, who will announce the result in early 2011.
We look forward to receiving your nominations.
Previous winners of the award include Professor Wilfred van der Donk, from University of Illinois, USA and Dr Akimitsu Okamoto, from RIKEN, Japan.
Comments Off on OBC Lecture Award 2011 – nominations now open!


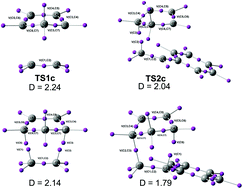









 Xianzhang Bu and colleagues at Sun Yat-Sen University in China have ‘unexpectedly’ discovered new fluorescent trans-dihydrofluoren-3-ones from one pot reactions of benzaldehydes and acetylacetone.
Xianzhang Bu and colleagues at Sun Yat-Sen University in China have ‘unexpectedly’ discovered new fluorescent trans-dihydrofluoren-3-ones from one pot reactions of benzaldehydes and acetylacetone.