The 10 most-accessed Green Chemistry articles between July and September 2013 were as follows:
Synthesis of thioesters through copper-catalyzed coupling of aldehydes with thiols in water
Chih-Lun Yi, Yu-Ting Huang and Chin-Fa Lee
Green Chem., 2013,15, 2476-2484, DOI: 10.1039/C3GC40946E, Paper
Highly efficient iron(0) nanoparticle-catalyzed hydrogenation in water in flow
Reuben Hudson, Go Hamasaka, Takao Osako, Yoichi M. A. Yamada, Chao-Jun Li, Yasuhiro Uozumi and Audrey Moores
Green Chem., 2013,15, 2141-2148, DOI: 10.1039/C3GC40789F, Paper
Iodine-mediated arylation of benzoxazoles with aldehydes
Yew Chin Teo, Siti Nurhanna Riduan and Yugen Zhang
Green Chem., 2013,15, 2365-2368, DOI: 10.1039/C3GC41027G, Communication
Polymer anchored Cu(II) complex: an efficient and recyclable catalytic system for the one-pot synthesis of 1,4-disubstituted 1,2,3-triazoles starting from anilines in water
Susmita Roy, Tanmay Chatterjee and Sk. Manirul Islam
Green Chem., 2013,15, 2532-2539, DOI: 10.1039/C3GC41114A, Paper
Multicomponent reactions in unconventional solvents: state of the art
Yanlong Gu
Green Chem., 2012,14, 2091-2128, DOI: 10.1039/C2GC35635J, Critical Review
Photocatalysis on supported gold and silver nanoparticles under ultraviolet and visible light irradiation
Sarina Sarina, Eric R. Waclawik and Huaiyong Zhu
Green Chem., 2013,15, 1814-1833, DOI: 10.1039/C3GC40450A, Tutorial Review
Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation
Jonathan G. Huddleston, Ann E. Visser, W. Matthew Reichert, Heather D. Willauer, Grant A. Broker and Robin D. Rogers
Green Chem., 2001,3, 156-164, DOI: 10.1039/B103275P, Paper
Deconstruction of lignocellulosic biomass with ionic liquids
Agnieszka Brandt, John Gräsvik, Jason P. Hallett and Tom Welton
Green Chem., 2013,15, 550-583, DOI: 10.1039/C2GC36364J, Critical Review
Hydrolysis of cellulose to glucose by solid acid catalysts
Yao-Bing Huang and Yao Fu
Green Chem., 2013,15, 1095-1111, DOI: 10.1039/C3GC40136G, Tutorial Review
Catalytic conversion of biomass to biofuels
David Martin Alonso, Jesse Q. Bond and James A. Dumesic
Green Chem., 2010,12, 1493-1513, DOI: 10.1039/C004654J, Critical Review
Take a look at the articles and then let us know your thoughts and comments below.
Fancy submitting your own work to Green Chemistry? You can submit online today, or email us with your ideas and suggestions.












 The front cover this month (pictured left) features a review by Jesse Hensley and co-workers from Golden, Colorado. In their article they focus on recent model compound studies of catalysts for hydrodeoxygenation of biomass pyrolysis products, with an emphasis on mechanisms, reaction networks, and structure–function relationships.
The front cover this month (pictured left) features a review by Jesse Hensley and co-workers from Golden, Colorado. In their article they focus on recent model compound studies of catalysts for hydrodeoxygenation of biomass pyrolysis products, with an emphasis on mechanisms, reaction networks, and structure–function relationships. The inside front cover this month (pictured right) features work by Andreas Heyden and co-workers from Columbia, South Carolina. In their work they report a theoretical study of the effects of various solvents on the mechanism of the hydrodeoxygenation of propanoic acid over Pd(111).
The inside front cover this month (pictured right) features work by Andreas Heyden and co-workers from Columbia, South Carolina. In their work they report a theoretical study of the effects of various solvents on the mechanism of the hydrodeoxygenation of propanoic acid over Pd(111).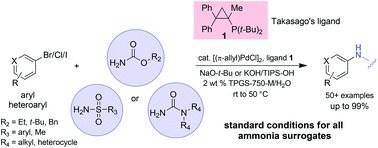
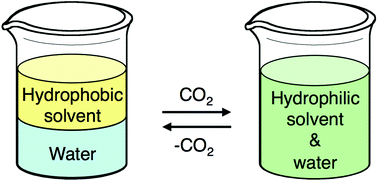
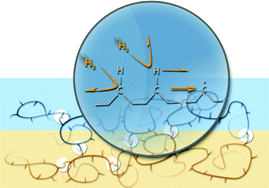
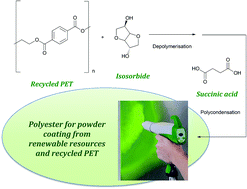
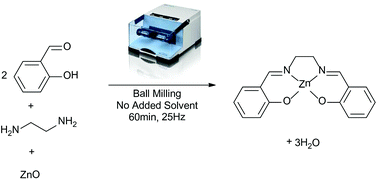
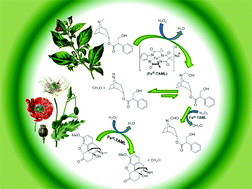
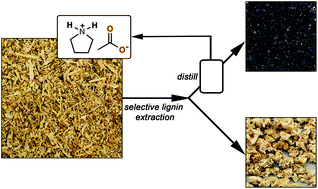

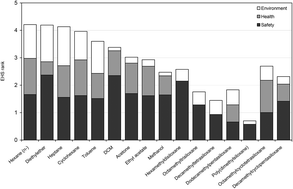
 The front cover this month (pictured left) features work by Brian Davison and co-workers. In their work they investigate the mechanism of biomass breakdown. Understanding this process should lead to more efficient use of biomass.
The front cover this month (pictured left) features work by Brian Davison and co-workers. In their work they investigate the mechanism of biomass breakdown. Understanding this process should lead to more efficient use of biomass. The inside front cover this month (pictured right) features work by Kevin Moeller and co-workers from Missouri, USA. In their work they set up a simple solar-electrochemical reaction to recycle Os(VIII)-, TEMPO-, Ce(IV)-, Pd(II)-, Ru(VIII)-, and Mn(V)-oxidants.
The inside front cover this month (pictured right) features work by Kevin Moeller and co-workers from Missouri, USA. In their work they set up a simple solar-electrochemical reaction to recycle Os(VIII)-, TEMPO-, Ce(IV)-, Pd(II)-, Ru(VIII)-, and Mn(V)-oxidants.
 The front cover this month (pictured left) features work by Peter C. K. Lau and co-workers from Quebec, Canada. In their work they engineer sinapic acid decarboxylaseas an alternative to chemistry-based or thermal decarboxylation to produce canolol from canola meal.
The front cover this month (pictured left) features work by Peter C. K. Lau and co-workers from Quebec, Canada. In their work they engineer sinapic acid decarboxylaseas an alternative to chemistry-based or thermal decarboxylation to produce canolol from canola meal. The inside front cover this month (pictured right) features work by Joerg Schrittwieser, Frank Hollmann and co-workers from Deltf, The Netherlands. In their work they show how the one-pot combination of alcohol dehydrogenase (ADH) and palladium nanoparticle (Pd-NP) catalysis provides access to aromatic 1,2-amino alcohols in high yields and excellent optical purities.
The inside front cover this month (pictured right) features work by Joerg Schrittwieser, Frank Hollmann and co-workers from Deltf, The Netherlands. In their work they show how the one-pot combination of alcohol dehydrogenase (ADH) and palladium nanoparticle (Pd-NP) catalysis provides access to aromatic 1,2-amino alcohols in high yields and excellent optical purities.