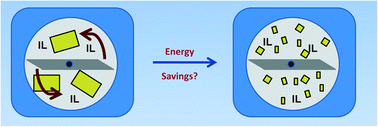
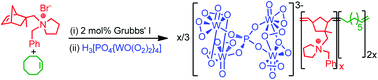
Scientists have developed an efficient peroxophosphotungstate-based polymer-immobilised ionic liquid phase catalyst for the epoxidation of allylic alcohols and alkenes.
Polyoxometalates (POMs) have been shown to be efficient Bronsted acid catalysts and selective oxidation catalysts in various important transformations. Research in recent years has been focused on surface immobilisation of POMs in order to develop continuous flow process and improve the robustness and recyclability of the catalysts and to avoid or reduce catalyst leaching.
This article is free to access until the 19th April 2012! Click on the link below to find out more…
An efficient recyclable peroxometalate-based polymer-immobilised ionic liquid phase (PIILP) catalyst for hydrogen peroxide-mediated oxidation, Simon Doherty, Julian G. Knight, Jack R. Ellison, David Weekes, Ross W. Harrington, Christopher Hardacre and Haresh Manyar, Green Chem., 2012, DOI: 10.1039/C2GC16679H