This month sees the following articles in Organic & Biomolecular Chemistry that are in the top ten most accessed:
Recent advances in indole syntheses: New routes for a classic target
Rubén Vicente
Org. Biomol. Chem., 2011, Advance Article
DOI: 10.1039/c1ob05750b
Bent bonds, the antiperiplanar hypothesis and the theory of resonance. A simple model to understand reactivity in organic chemistry
Ghislain Deslongchamps and Pierre Deslongchamps
Org. Biomol. Chem., 2011, 9, 5321-5333
DOI: 10.1039/c1ob05393k
The nucleophilicity N index in organic chemistry
Luis R. Domingo and Patricia Pérez
Org. Biomol. Chem., 2011, Advance Article
DOI: 10.1039/c1ob05856h
First asymmetric cascade reaction catalysed by chiral primary aminoalcohols
Carlos Arróniz, Carmen Escolano, F. Javier Luque, Joan Bosch and Mercedes Amat
Org. Biomol. Chem., 2011, 9, 5079-5085
DOI: 10.1039/c1ob05400g
A synergistic approach to polycyclics via a strategic utilization of Claisen rearrangement and olefin metathesis
Sambasivarao Kotha, Nimita G. Krishna, Somnath Halder and Shilpi Misra
Org. Biomol. Chem., 2011, 9, 5597-5624
DOI: 10.1039/c1ob05413a
Kopsihainanines A and B, two unusual alkaloids from Kopsia hainanensis
Jia Chen, Jian-Jun Chen, Xiaojun Yao and Kun Gao
Org. Biomol. Chem., 2011, 9, 5334-5336
DOI: 10.1039/C1OB05724C
Aromatic C-nitrosation of a bioactive molecule. Nitrosation of minoxidil
Mario González-Jiménez, Jorge Arenas-Valgañón, Emilio Calle and Julio Casado
Org. Biomol. Chem., 2011, Advance Article
DOI: 10.1039/c1ob05686g
Selection of the biological activity of DNJ neoglycoconjugates through click length variation of the side chain
Nicolas Ardes-Guisot, Dominic S. Alonzi, Gabriele Reinkensmeier, Terry D. Butters, Caroline Norez, Frédéric Becq, Yousuke Shimada, Shinpei Nakagawa, Atsushi Kato, Yves Blériot, Matthieu Sollogoub and Boris Vauzeilles
Org. Biomol. Chem., 2011, 9, 5373-5388
DOI: 10.1039/c1ob05119a
An organocatalytic ionic liquid
Zsolt Kelemen, Oldamur Hollóczki, József Nagy and László Nyulászi
Org. Biomol. Chem., 2011, 9, 5362-5364
DOI: 10.1039/C1OB05639E
Mild preparation of functionalized [2.2]paracyclophanes via the Pummerer rearrangement
Matteo Montanari, Alberto Bugana, Arvind K. Sharma and Dario Pasini
Org. Biomol. Chem., 2011, 9, 5015-5020
DOI: 10.1039/c1ob05319a
Why not take a look at the articles today and blog your thoughts and comments below.
Fancy submitting an article to Organic & Biomolecular Chemistry? Then why not submit to us today or alternatively email us your suggestions.
Comments Off on Top ten most accessed articles in July
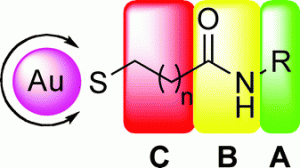 Sabine Schlecht and colleagues from the Free University of Berlin have designed a series of sulfated cyclic and acyclic amino alcohols bound to colloidal gold nanoparticles for use as multivalent selectin binding agents. The carbohydrate mimics were chosen to act as simplified analogs of the sLex unit found in biological selectin ligands, and demonstrated extremely high binding affinities towards L- and P-selectin and no cytotoxicity.
Sabine Schlecht and colleagues from the Free University of Berlin have designed a series of sulfated cyclic and acyclic amino alcohols bound to colloidal gold nanoparticles for use as multivalent selectin binding agents. The carbohydrate mimics were chosen to act as simplified analogs of the sLex unit found in biological selectin ligands, and demonstrated extremely high binding affinities towards L- and P-selectin and no cytotoxicity.










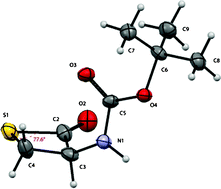
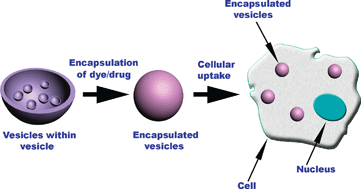
![CoverIssue[4]](https://blogs.rsc.org/ob/files/2011/09/CoverIssue4.gif)
![GA[6]](https://blogs.rsc.org/ob/files/2011/09/GA6.gif)

![GA[4]](https://blogs.rsc.org/ob/files/2011/08/GA4.gif)
