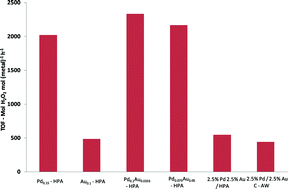
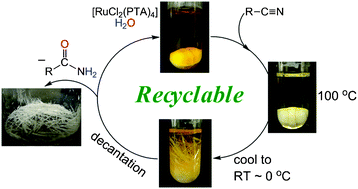
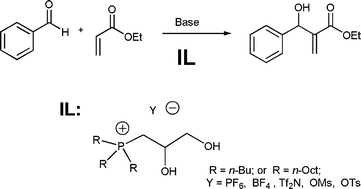
This month sees the following articles in Green Chemistry that are in the top ten most accessed:-
Recent advances in ionic liquid catalysis
Qinghua Zhang, Shiguo Zhang and Youquan Deng
Green Chem., 2011, Advance Article, DOI: 10.1039/C1GC15334J
Green synthesis of metal nanoparticles using plants
Siavash Iravani
Green Chem., 2011, Advance Article, DOI: 10.1039/C1GC15386B
Functional ionic liquid mediated synthesis (FILMS) of dihydrothiophenes and tacrine derivatives
Atul Kumar, Garima Gupta and Suman Srivastava
Green Chem., 2011, 13, 2459-2463, DOI: 10.1039/C1GC15410A
Enzymatic reductions for the chemist
Frank Hollmann, Isabel W. C. E. Arends and Dirk Holtmann
Green Chem., 2011, 13, 2285-2314, DOI: 10.1039/C1GC15424A
Sustainable hydrogen production by the application of ambient temperature photocatalysis
Michael Bowker
Green Chem., 2011, 13, 2235-2246, DOI: 10.1039/C1GC00022E
Could the energy cost of using supercritical fluids be mitigated by using CO2 from carbon capture and storage (CCS)?
James G. Stevens, Pilar Gómez, Richard A. Bourne, Trevor C. Drage, Michael W. George and Martyn Poliakoff
Green Chem., 2011, 13, 2727-2733, DOI: 10.1039/C1GC15503B
One step catalytic conversion of cellulose to sustainable chemicals utilizing cooperative ionic liquid pairs
Jinxing Long, Bin Guo, Xuehui Li, Yanbin Jiang, Furong Wang, Shik Chi Tsang, Lefu Wang and Kai Man K. Yu
Green Chem., 2011, 13, 2334-2338, DOI: 10.1039/C1GC15597K
Ionic liquid pretreatment of lignocellulosic biomass with ionic liquid–water mixtures
Agnieszka Brandt, Michael J. Ray, Trang Q. To, David J. Leak, Richard J. Murphy and Tom Welton
Green Chem., 2011, 13, 2489-2499, DOI: 10.1039/C1GC15374A
The solvent-free and catalyst-free conversion of an aziridine to an oxazolidinone using only carbon dioxide
Chau Phung, Rani M. Ulrich, Mostafa Ibrahim, Nathaniel T. G. Tighe, Deborah L. Lieberman and Allan R. Pinhas
Green Chem., 2011, Advance Article, DOI: 10.1039/C1GC15850C
Microwave assisted conversion of carbohydrates and biopolymers to 5-hydroxymethylfurfural with aluminium chloride catalyst in water
Sudipta De, Saikat Dutta and Basudeb Saha
Green Chem., 2011, 13, 2859-2868, DOI: 10.1039/C1GC15550D
Why not take a look at the articles today and blog your thoughts and comments below.
Fancy submitting an article to Green Chemistry? Then why not submit to us today or alternatively email us your suggestions.
Comments Off on Top ten most accessed articles in September