A new porous co-ordination polymer has been synthesised in near-critical water which could broaden the synthetic strategies available for the environmentally friendly construction of metal-organic frameworks.
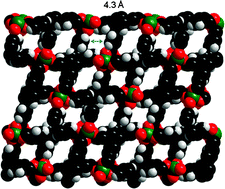
In this work Martyn Poliakoff and Martin Schröder and colleagues from the University of Nottingham and Diamond Light Source, UK, have developed a route to synthesise these Zn co-ordination polymers using near-critical water (300°C) as a cleaner alternative to organic solvents. The zinc complex can be isolated without any water molecules bound to the Zn(II) centres, in contrast to solvothermal synthesis in DMF. In addition, the use of water in this synthetic strategy potentially allows for the reuse and recovery of water from the reaction opening up possible industrial applications.
This article is free to access until the 20th December 2011! Click on the link below to find out more…
Near-critical water, a cleaner solvent for the synthesis of a metal–organic framework, Ilich A. Ibarra, Peter A. Bayliss, Eduardo Pérez, Sihai Yang, Alexander J. Blake, Harriott Nowell, David R. Allan, Martyn Poliakoff and Martin Schröder, Green Chem., 2012, DOI: 10.1039/C1GC15726D