From June 18th to 20th, 2013, the Cluster of Excellence “Tailor-Made Fuels From Biomass” (TMFB), which is based at RWTH Aachen University organized its 1st International Conference in Aachen. The conference was held in the host city of Aachen, which is located at the Vaalserberg triple point between Germany, the Netherlands and Belgium and was made famous by Charlemagne. The three-day conference covered all subject areas surrounding the synthesis, production and combustion of modern biofuels and addressed an international, interdisciplinary audience from academia and industry. Since 2007, researchers from the fields of chemistry, biology, process engineering, mechanical engineering and associated research fields have been working together on the development of tailored biofuels within the TMFB. In previous years, the Cluster of Excellence used its “International Workshop” as a platform for the exchange of TMFB Members with external researchers. This year the conference’s stage was opened, and for the first time more than half of the 40 speakers visited from external institutions. The program included lectures from invited speakers and researchers from inside and outside the Cluster of Excellence as well as a poster session, where young researchers from different scientific disciplines were able to present and discuss their work.
The first day of the conference emphasized the interdisciplinary approach of the TMFB by combining together lectures addressing the interaction of the involved disciplines, namely the transformation of lignocellulose into biofuels, the combustion properties of alternative fuels, the importance of lubrication in combustion engines and the health concerns associated with exhaust gases. Bringing together all of the conference participants for this series of diverse lectures highlighted the importance of examining the development of the next generation of biofuels from all perspectives.

Prof. Tom Welton presenting on Ionic Liquids for Biomass Fractionation
The lecture portion of the conference began, according to the value chain of biofuel production, with the fractionation of lignocellulose. Prof. Tom Welton outlined how the unique environment provided by ionic liquids can solubilize lignocellulosic feedstocks, which generally have low solubilities in aqueous and organic solutions. By applying a novel pretreatment method called Ionosolv, 80% of the lignin can be separated without changing the native structure of the lignin molecules. However, Prof. Welton pointed out that the obtained cellulose fraction has to be washed carefully to avoid the inactivation of cellulases by small amounts of ionic liquid. The dissolution of lignocellulose in ionic liquids allowed for the separation of lignin, cellulose and hemicellulose, such that the cation and anion of the ionic liquid could be tailored to improve the separation of authentic lignocellulose materials.

The TMFB lego model, explaining the TMFB biofuel production approach
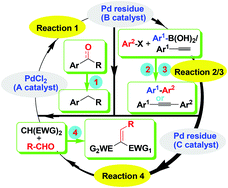
Two members of TMFB, Prof. Jürgen Klankermayer (Technical Chemistry) and Manuel Dahmen (Process Technology, Mechanical Engineering), presented on the collaborative approach being developed within the Cluster of Excellence to define and synthesize biofuel targets. Mr. Dahmen outlined the development of theoretical models to identify potential biofuels based on predicting the combustion fuel properties (i.e. boiling point, heating value, cetane number) of biomass-derived substrates. The insight provided by this predictive property model identifies which molecular structures have suitable fuel properties and helps to direct the synthetic efforts within the TMFB cluster. Prof. Klankermayer highlighted the various catalytic strategies, both homogeneous and heterogeneous, being developed within the TMFB cluster toward the synthesis of the next generation of biofuels including 2-methyltetrahydrofuran and 1-octanol. The combined presentation from two lecturers of two different disciplines at the same time formed a vital example of the successfully implemented interdisciplinary research culture in Aachen’s Cluster of Excellence.
Prof. Paul Anastas, the father of Green Chemistry, presented the closing seminar on the first day of the conference. Rather than discussing his exceptional research work on such topics as the theoretical prediction of chemical toxicity or the integration of process design into Green Chemistry practices, Prof. Anastas presented his philosophy on the current state of Green Chemistry. His stimulating and motivating presentation illustrated the importance of Green Chemistry by providing numerous examples on the implementation of green technologies and challenged the conference participants to continue to strive in the development of a sustainability chemical industry by creating truly innovative technologies. He addressed several questions regarding the establishment of green chemistry and its impact on today’s society, leading to many excited discussions at the get-together in the evening.
On the second day of the conference, Prof. Charles Westbrook outlined his work on the development of kinetic models to explain the ignition and combustion of alternative fuels. His lecture provided evidence on the relationship between combustion properties and the molecular structure of a biofuel, which again illustrated how combustion models can help to guide synthetic chemists towards optimal molecular structures. Furthermore, his work emphasized how several combustion techniques must be combined to provide a suitable model and as such requires interdisciplinary collaboration to provide answers to this sustainability problem.
The remainder of the second day of the conference was filled with excellent lectures provided by both internal and external speakers on a variety of chemistry, biology and engineering topics. The afternoon presentations and discussions identified that lignocellulose pretreatment methods and the characterization of the resulting biomass fractions are vital to all downstream processes. Qingqi Yan presented a way to pretreat the biomass mechanically using a screw press. The advantages of this device are the universal usage for all kinds of lignocellulosic biomass, the scalability, the robustness, the low operational costs and the high disruption of lignocellulosic structures. By combining the screw press with the chemical hydrolysis in one step, valuable reducing sugars can be produced in a very early stage of the process chain. Furthermore, Prof. Markus Pauly presented on the use of 2D NMR spectroscopy for the characterization of lignocellulosic materials. The identification of lignocellulose presents a particular problem to chemists due to its complex structure and low solubility. Prof. Pauly outlined a technique to solubilize lignocellulose using deuterated ionic liquids and dimethyl sulfoxide and analyze the various components of this biopolymer using 2D NMR. This technique has provided a method to investigate the influence of various plant feedstocks on the structure of lignocellulose and to evaluate different methods used in the fractionation of lignocellulose.
The Combustion Kinetics session covered various aspects of biofuel kinetics and emphasized the paramount importance of international collaboration in this field. John Dec started the session by giving detailed insight into the differences of the reaction chemistry of bio-ketones as compared to conventional fuels and also discussed the implications of these differences to novel combustion concepts. His talk covered the development of the reaction mechanism for di-n-butylether (DNBE) as a TMFB example. Further, his talk emphasized the statement made by Prof. Westbrook during the morning’s invited lecture, that such mechanism development is the work of a complete “gang” of scientists, bringing together the enormous experimental and brain power needed worldwide.
The Fuels Spray, Flow and Mixing session covered physicochemical effects on the combustion system as well as experimental visualizations of biofuel injection compared to and complemented by high-fidelity simulations. The session closed with a study on injector deposits resulting from the applications ofbiofuels presented by Dr. Christian Fink from Rostock University. In the Combustion Systems Session, a strong focus was on Diesel engine application of various alternative fuels and fuel blends. Prof. Eilts from the Technical University Braunschweig, pointed at the potential of higher alcohols to reduce soot when blended with Diesel fuel. Unfortunately, adverse effects on the number of very small particles were also reported. A more detailed study on fuel effects in Diesel engine combustion was reported for a comparison of DNBE, octanol and Diesel. Combined examinations on single cylinder diesel engine test bench, high pressure spray chamber and CFD simulations of both experiments reveal that to reach clean combustion, the mixing behavior may be more important than the cetane rating. The very good mixture preparation obtained with DNBE enables soot-free low-NOx combustion for the entire engine load range, despite the high cetane rating (about 100) of DNBE.

Discussions during the Poster Session at the 1st TMFB International Conference
Another session in the afternoon covered the pathways from biofuels to propulsion. Well to wheel balances of blends confirming to EN 590 surveyed sustainability aspects of already usable fuels. The closing talk, presented by Prof. Claus Felby from the University of Copenhagen, dealt with the question of bringing bio-refineries to large scale, which will be a key factor with regards to cost and also energy efficiency of biofuel processes.
To analyze the biomass composition with respect to its glucose, xylose and cellulose content, Helene Wulfhorst from the University of Kaiserslautern showed the potential and challenges of applying near- and mid-infrared spectroscopy. To exploit the possibilities of this technique, choosing the optimal calibration strategy is absolutely essential. For this reason different calibration strategies were compared with each other, to demonstrate the best way of analyzing biomass composition. An essential step to convert biomass into a biofuel or platform chemical is fermentation. Irina Borodina, a researcher from the Danish Chalmers University of Technology, made a short digression to the biological corner of the conference by presenting the potential of yeast cells in general as cell factories for biorefinery processes. The fact that yeasts are not the only opportunities to convert biomass was shown by the three presentations of Jasmine Roth from the University of Kaiserslautern, Sandra Wewetzer/Frederike Carstensen from the RWTH Aachen University and Simon Curvers from Direvo Industrial Biotechnology GmbH. Jasmine Roth presented experimental and modeled data of acetone-butanol-ethanol (ABE) fermentations based on Clostridium acetobutylicum and complex liquid hydrolysates combined with an in situ product removal by particle-based extraction. Sandra Wewetzer and Frederike Carstensen introduced a process concept for the conversion of glucose into itaconic acid using the fungus Ustilago maydis. To increase the efficiency of the process a membrane module is integrated into the bioreactor to enable an in situ product recovery. On the last conference day Simon Curvers presented the BlueCon® concept of the company Direvo which allows for the direct conversion of a broad range of substrates into ethanol and / or lactic acid. The fermentations were performed with the extremely thermophilic Caldicellulosiruptor strains without any additionally added enzymes.
Prof. Julie Zimmerman was invited to present her work towards the development of algae for the production of biofuels. Her presentation outlined how this important biological resource can be grown, separated into its different components and catalytically transformed into biofuels. Her work illustrated how process integration employing supercritical carbon dioxide can permit the fractionation of algae and transesterification within a single reactor to reduce the energy costs of harvesting this resource for fuels and chemicals. Her work combines together biochemistry and engineering to provide another excellent example of the interdisciplinary approach required to tackle sustainability issues.
Apart from the interesting scientific lectures and discussions at day, there was also a supporting program in the evenings. On the first evening, a get-together with Bavarian-style food and drinks was organized to create the opportunity to network with new acquaintances and catch up with established ones. On the second evening, all lecturers, professors, principal investigators, external guests and selected additional TMFB members were invited to the Conference Dinner in the Lenné-pavillon at the Casino Aachen, which is made of glass and offers a beautiful view of the fountain in the Kurpark. With the sunny weather, the terrace of the pavillon was the place where the great part of the dinner guests came together to eat, drink and enjoy the pleasant and inspiring conversations.
In conclusion, the 1st International Conference of the Cluster of Excellence TMFB has set the bar high for the second TMFB conference, which will take place again in Aachen, June 16th to 18th, 2014. Researchers interested in interdisciplinary perspectives on new biofuels are most likely to benefit once more from the valuable input, animated discussions and great networking opportunities.
![GA[2]](https://blogs.rsc.org/gc/files/2013/10/GA2.gif)












![GA[3]](https://blogs.rsc.org/gc/files/2013/09/GA3.gif)

![GA[1]](https://blogs.rsc.org/gc/files/2013/09/GA1.gif)
![GA[2]](https://blogs.rsc.org/gc/files/2013/09/GA2.gif)