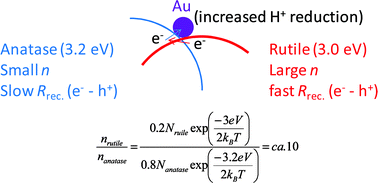
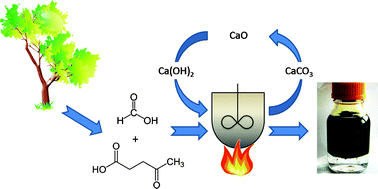
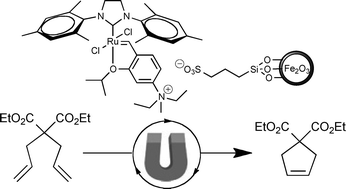
In this Perspective article, William Watson, Scientific Update LLP, UK, looks at the approach of companies within the chemical, pharmaceutical and related industries to green chemistry and sustainability, and examines how much variation exists from company to company.
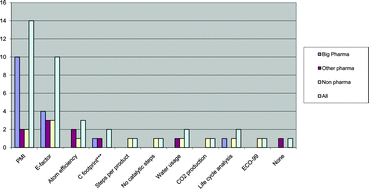
The study approached several companies from across the sector and included 11 ‘big pharma’ companies, 6 ‘other pharma’ companies and 4 non-phrama companies who agreed to take part in the survey. The survey itself looked at green chemistry and sustainability policies and policy implementation, process metrics, green chemistry and technologies and how the application of the green chemistry principles changes throughout the various stages of development.
The study highlights that the overall picture across the sector is very varied, with a few companies clearly leading the field and some slower to introduce or highlight green issues than others. However, all companies try to use green solvents where possible and avoid using solvents like dichloromethane.
This article is now free to access until the 3rd February 2012! Click the link below to find out more…
How do the fine chemical, pharmaceutical, and related industries approach green chemistry and sustainability?, William J. W. Watson, Green Chem., 2012, DOI: 10.1039/C1GC15904F











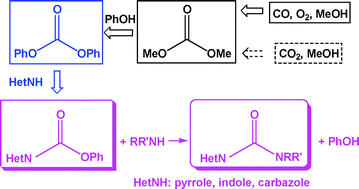
 Anett Schallmey and Bruno Bühler,
Anett Schallmey and Bruno Bühler, 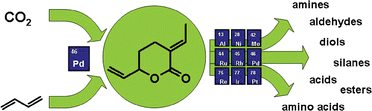 Use of carbon dioxide in chemical syntheses via a lactone intermediate
Use of carbon dioxide in chemical syntheses via a lactone intermediate