Molecular self-assembly has become one of the most prominent fields of nanotechnology in recent years. Inspired by nature, many scientists around the world are attempting to utilize it as a tool to design novel nanostructures with desired biomedical properties.
To achieve this goal, it is necessary to understand how self-assembly works and how molecular forces and other conditions drive this self-assembly and define the structure of a specific supramolecular complex.
In a recent Organic & Biomolecular Chemistry publication, Professor Charlotte J. C. Edwards-Gayle and Professor Ian W. Hamley from the University of Reading, UK, have reviewed some of the most prominent self-assembled peptide amphiphiles and their potential applications. These include tissue scaffolds, antimicrobial peptides and drug transporters.
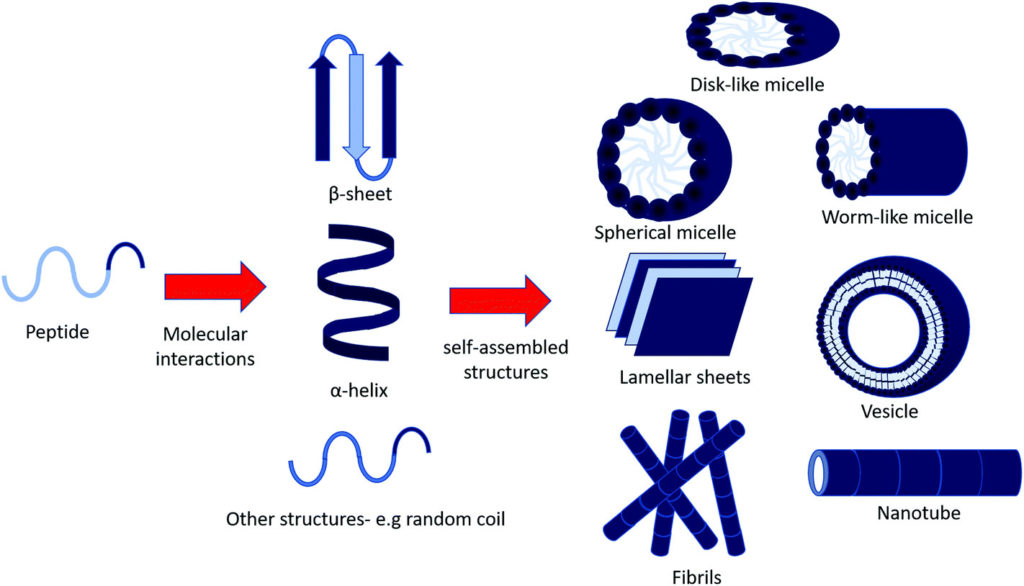
One of the distinct features of self-assembling amphiphiles, which makes them attractive candidates for many applications, is their diversity to form various structures such as micelles, vesicles, nanotubes, fibrils and sheets (Fig.1). The self-assembly of PAs can also be tuned by several factors including amino acid sequence, peptide length, temperature, pH, and concentration.
These features make them perfect, novel tools to create biomaterials which can be responsive to different environmental cues. However, the review points out that the key question of whether there is a relationship between bioactivity and self-assembly of peptide amphiphiles has still remained unanswered, despite the advancement in the field.
Extending the success in designing various structures, together with distinct applications, have made peptide amphiphile self-assembly a vibrant field in which researchers will continue to develop functional constructs with novel applications.
To find out more please see:
Self-assembly of bioactive peptides, peptide conjugates, and peptide mimetic materials
Charlotte J. C. Edwards-Gayle and Ian W. Hamley
DOI: 10.1039/C7OB01092C
Zahra Bahrami Dizicheh is a PhD candidate in molecular biology with Dr. Giovanna Ghirlanda at Arizona State University. She does research on photo-electrochemical hydrogen production with designing and developing interconnection between conductive materials and redox proteins to develop dye-sensitized photo-electrochemical cells.