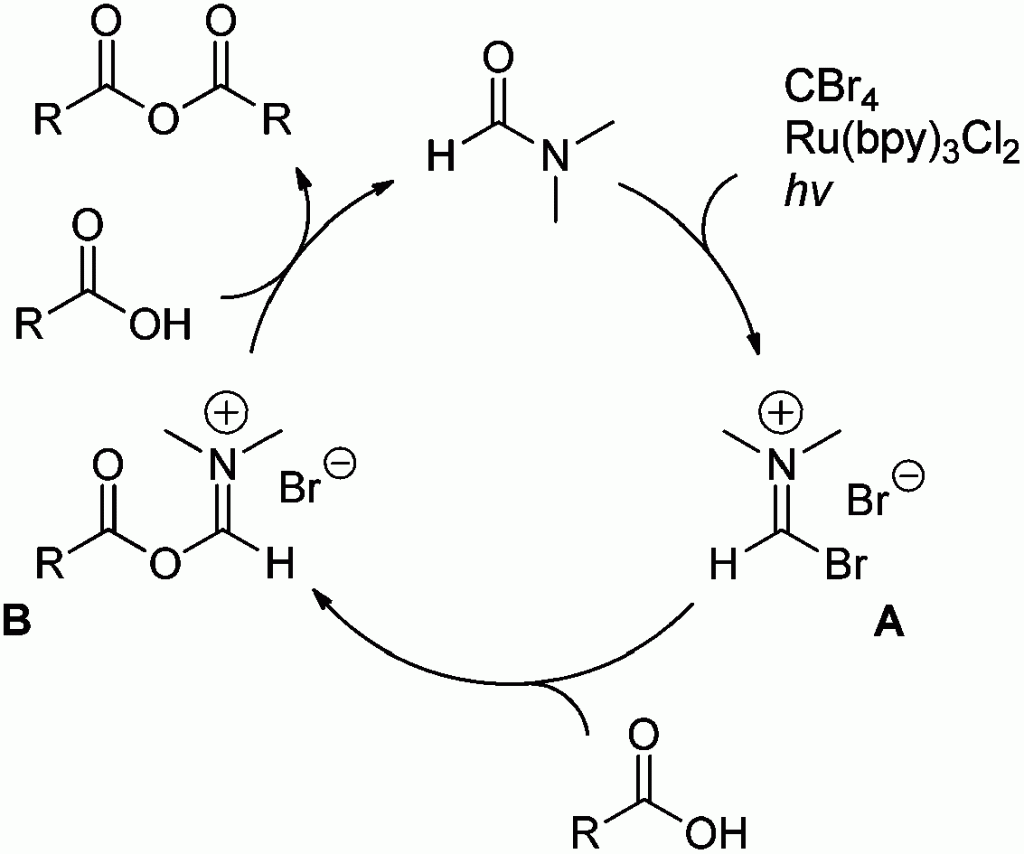
 In this HOT communication Corey R. J. Stephenson and colleagues at Boston University demonstrate the application of visible light-mediated photoredox catalysis for the synthesis of symmetric anhydrides. This new approach to anhydride formation proceeds via the activation of C–O bonds by the Vilsmeier–Haack reagent formed by Ru(bpy)3Cl2 and CBr4 in DMF.
In this HOT communication Corey R. J. Stephenson and colleagues at Boston University demonstrate the application of visible light-mediated photoredox catalysis for the synthesis of symmetric anhydrides. This new approach to anhydride formation proceeds via the activation of C–O bonds by the Vilsmeier–Haack reagent formed by Ru(bpy)3Cl2 and CBr4 in DMF.
Stephenson et al. demonstrate the reaction with various aryl and alkyl carboxylic acids being converted to the corresponding anhydrides in excellent yields and they go on to show the efficacy of the method by performing scale-up reactions.
For the full reaction conditions have a look at this Communication today. As always, this HOT article will be free to access for the next 4 weeks!
Synthesis of symmetric anhydrides using visible light-mediated photoredox catalysis
Marlena D. Konieczynska , Chunhui Dai and Corey R. J. Stephenson
DOI: 10.1039/C2OB25463H











 Welcome to OBC issue 22
Welcome to OBC issue 22







![GA[5]](https://blogs.rsc.org/ob/files/2012/05/GA5-300x141.gif)


