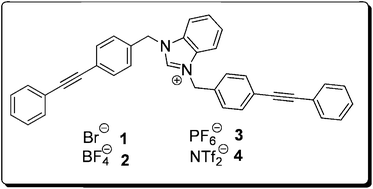
Its bonds are constantly rearranging through a seemingly endless series of Cope rearrangements. It’s estimated that there are over 1.2 million possible valence tautomers, and due to the rapidity of the conversions, all carbons and protons appear as equivalent on the NMR timescale.
Professor Jeff Bode and his group at ETH Zürich have an interest in these fluxional molecules and their potential uses as chemical sensors.
During a recent investigation into the racemisation of oligo-substituted members of the bullvalene family, they noticed some interesting behaviour in a sample of a tetrasubstituted bullvalene. A single isomer was spontaneously forming, and could be isolated from the mixture. This isomer was surprisingly stable, with a half-life of just over 4 hours at room temperature.
Now, tetrasubstituted bullvalenes have 1680 possible structural isomers and previous work has shown that the mixture is fully dynamic, so the appearance of just one isomer above all the others is pretty remarkable.
Their latest HOT PAPER builds on these results, and seeks to provide further information about the nature of this fascinating molecule.











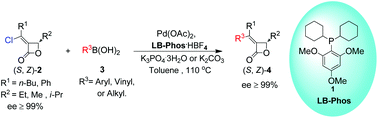
![Synthesis of a four-component [3]catenane using three distinct noncovalent interactions](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C2OB26587G)




![1,5-(H, RO, RS) shift/6π-electrocyclic ring closure tandem processes on N-[(α-heterosubstituted)-2-tolyl]ketenimines](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C2OB27010B)