Currently, nanomaterials (NM) are attracting significant attention in the field of biomedicine. However, once these nanomaterials are utilized for in vivo treatments they interact with the surrounding physiological environment, leading to the adsorption of various biomolecules onto their surfaces, forming a biomolecular corona (BMC) and thereby influencing the performance and behavior of the nanomaterials. Presently, the in vitro studies of NM primarily involve dispersing the nanoparticles in 10% fetal bovine serum (FBS) and then evaluating their toxicity and therapeutic effects. However, this evaluation method is insufficient as it cannot accurately simulate the conditions of human blood. Moreover, this practical issue remains unresolved to date.
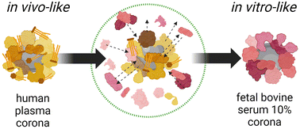
Fig 1. Schematic illustrating the molecular and biological biases arising from the well-known in vitro/in vivo mismatch in nanomedicine due to the biomolecular corona. Reproduced from DOI: 10.1039/D3NH00510K with permission from the Royal Society of Chemistry.
To validate the series of biases existing in established experimental practices and to advance the fields of nanomedicine and nanotoxicology, this study investigated two NM types with vastly different physicochemical properties commonly used in biomedicine. The research compared the molecular and biological biases resulting from the mismatch between NM dispersed in 10% FBS (utilized for in vitro biological assays) and whole human plasma (HP, closer to in vivo administration schemes). Through comparative analysis using proteomics, lipidomics, high-throughput multi-parameter in vitro screening, and single-molecule feature analysis, it was demonstrated that the dynamic changes in BMC composition are material dependent and that cell viability, transport pathways, and autophagic cascades are influenced by the presence or absence of pre-formed BMC corona. These findings underscore the potential limitations of NM in vitro testing in accurately representing real in vivo conditions. Therefore, it is necessary to establish new shared protocols to enhance the accuracy and predictive capability of NM testing.
In summary, this study confirms the biases that may exist when using standard in vitro conditions for NM toxicology assessments, reminding us of the need to establish a comprehensive experimental framework to generate and support new knowledge in the field of biologically relevant nanomaterial interactions. For instance, integrating advanced predictive tools such as artificial intelligence and machine learning will enable nanotoxicology and nanomedicine to progress towards personalized solutions for precision healthcare.
To find out more, please read:
Sources of biases in the in vitro testing of nanomaterials: the role of the biomolecular corona
Valentina Castagnola, Valeria Tomati, Luca Boselli, Clarissa Braccia, Sergio Decherchi, Pier Paolo Pompa, Nicoletta Pedemonte, Fabio Benfenati and Andrea Armirotti
Nanoscale Horiz., 2024, Advance Article
About the blogger

Fangfang Cao is a Research Fellow at National University of Singapore and a member of the Nanoscale Horizons Community Board. Dr Cao’s research focuses on nanocatalytic medicine and microbial therapy. |