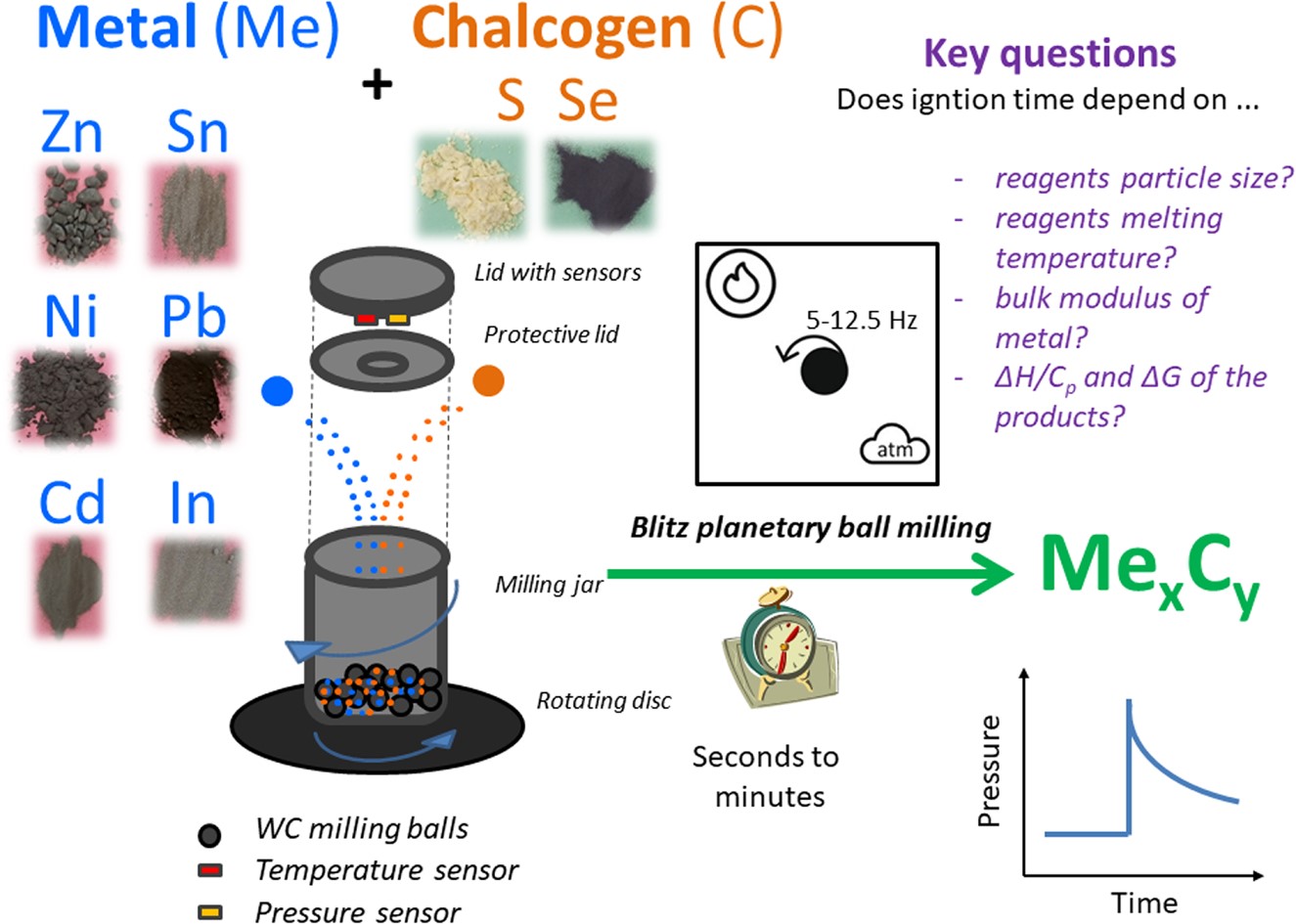
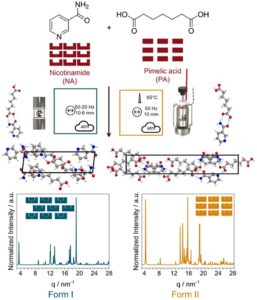
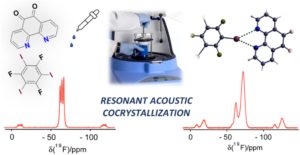
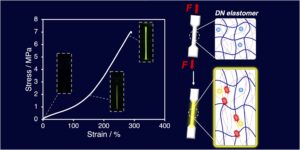
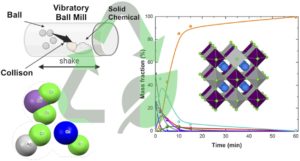
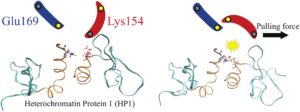
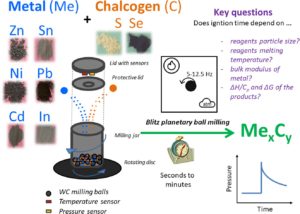
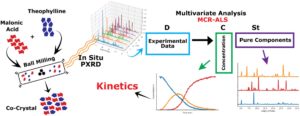
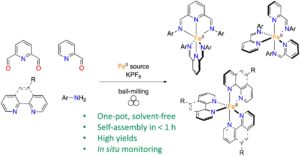
RSC Mechanochemistry has published its first articles. To celebrate this, we asked the authors to discuss their work in some more detail.
In this edition, we hear from Masad Damha, James Thorpe and Julian Marlyn about their study titled Synthesis of short DNA and RNA fragments by resonant acoustic mixing (RAM).
Want to know more about their work? Read the full paper here!
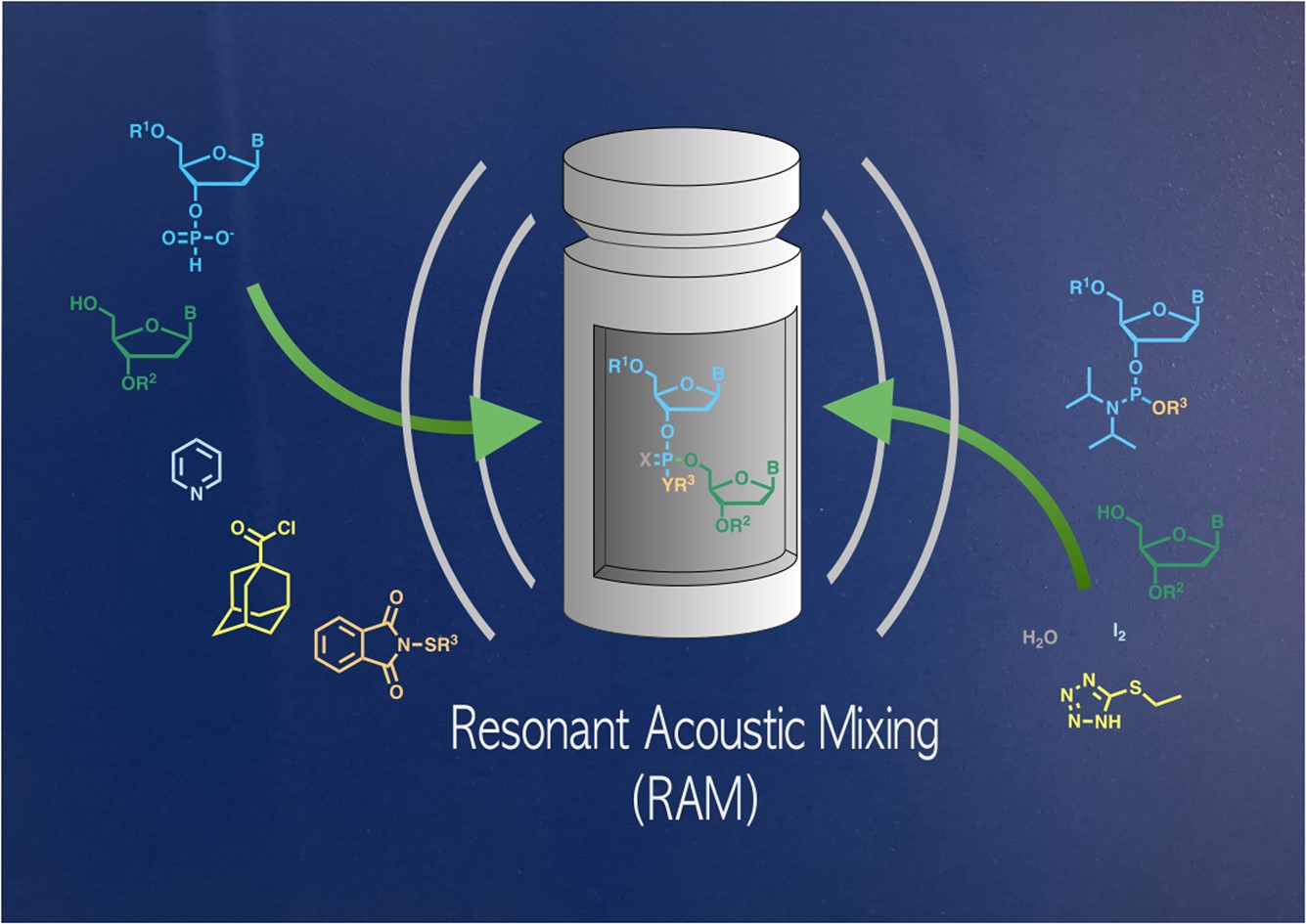
Synthesis of short DNA and RNA fragments by resonant acoustic mixing (RAM)
James D. Thorpe, Julian Marlyn, Stefan G. Koenig and Masad J. Damha
RSC Mechanochem. 2024, Advance Article, DOI: 10.1039/D4MR00009A
 |
RSC Mechanochemistry offers you an inclusive and dedicated home for the ideas, scientific language and approaches that cut across the many disciplines mechanochemistry touches. Here we are seeking to build knowledge, as well as foster innovation and discovery at this forefront of chemistry. Whether you are seeking to understand the fundamentals of mechanochemistry, or you are excited by its applications and potential, this journal is for you.
|