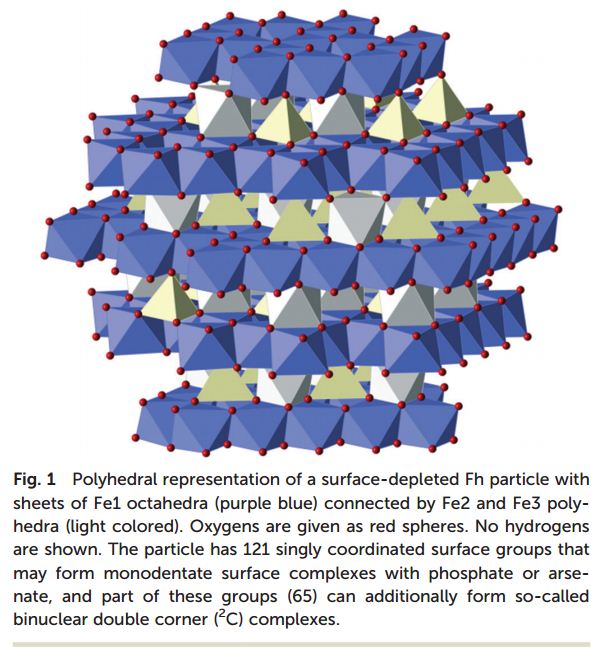
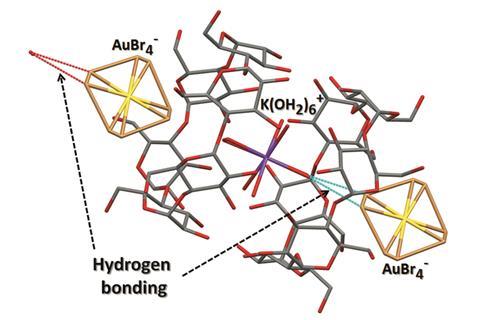
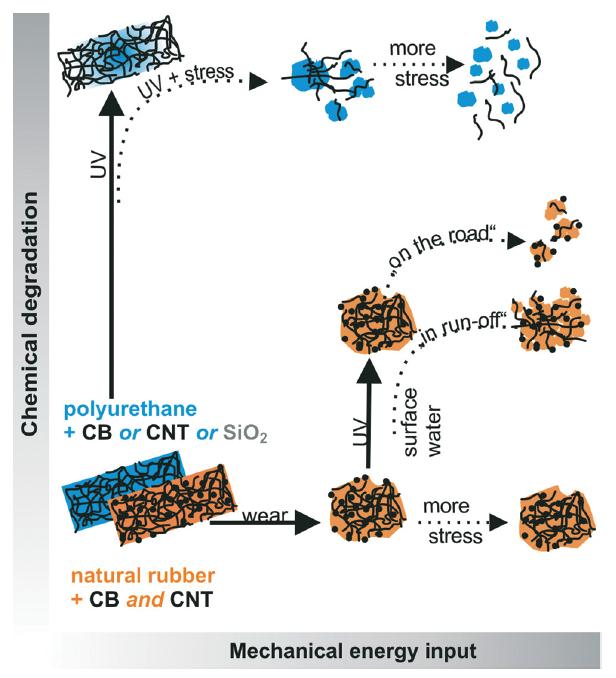
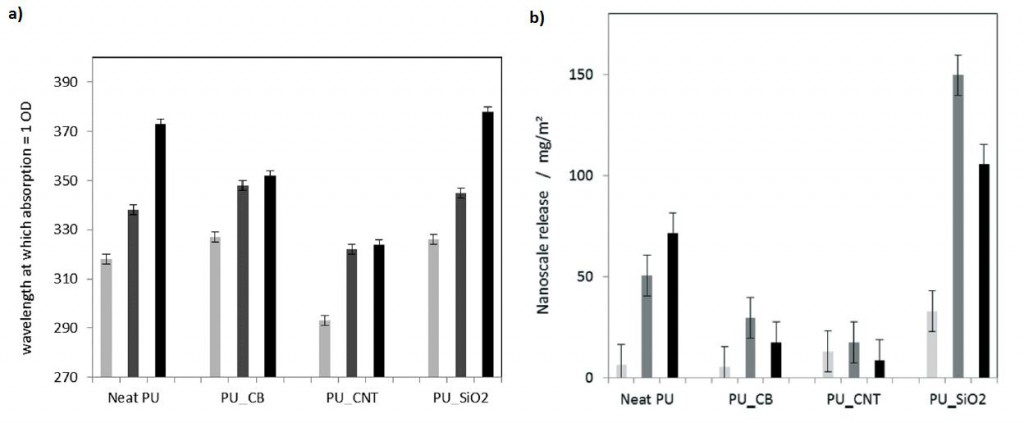
We are delighted to announce the appointment of the following people to the Environmental Science: Nano Advisory Board.
 |
Melanie Auffan
Melanie Auffan is a CNRS research scientist at the CEREGE (European Geosciences Center) in Aix en Provence. She is member of the iCEINT and CEINT steering committee (consortium for the Environmental Implications of Nanotechnology). Her research addresses the physico-chemical properties and surface reactivity of nanoparticles in contact with living organisms. |
 |
Yoon-Seok Chang
Professor Yoon-Seok Chang is based at Pohang University of Science and Technology (Postech), South Korea. His research interests include zero valent iron based nanotechnologies for groundwater remediation and the environmental fates and human health effects of toxic substances and nanomaterials. |
 |
Philip Demokritou
Dr Demokritou is currently an Associate Professor at Harvard School of Public Health. His research interests are primarily in the areas of nano-aerosol science and technology with emphasis on the elucidation of particle health effects.
|

|
Juliane Filser
Juliane Filser is a full professor for general and theoretical ecology and vice director of the interdisciplinary UFT Center for Environmental Research and Sustainable Technology at the University of Bremen, Germany. Juliane’s main research focus is prospective environmental risk assessment, with special attention to ecological interactions in soils. Her group had been one of the first worldwide to point out the need for assessing potential risks of nanoparticles in terrestrial environments. |
 |
John Fortner
Professor Fortner’s research is primarily focused on environmental implications and applications of advanced materials. He has extensively studied the environmental fate, reactivity and impacts of engineered carbon nanomaterials, including fullerenes and carbon nanotubes, in aqueous systems.
|

|
Robert Hurt
Robert H. Hurt is Professor of Engineering at Brown University, USA. His current research includes the biological response to graphene-family nanomaterials, mechanisms of carbon nanotube uptake and toxicity, nano-silver and nano-copper transformations in the natural environment, safe material design, and the assembly and folding of graphene to make three-dimensional architectures for barrier and encapsulation technologies, and as electrodes and catalyst supports. |

|
Saber Hussain
Saber Hussain is Senior Scientist and Nanotoxicology Group Lead, Molecular Bioeffects Division, Wright-Patterson Air Force Base, Ohio. His research interests focus on the fundamental interaction of engineered nanomaterials with biological systems, with a special focus on developing nanodevices and evaluating potential toxicity arising from the physicochemical properties of nanoscale structures. |
 |
Ralf Kaegi
Dr Ralf Kaegi is based at EAWAG, Switzerland. His research interests are focused on the fate and transport of engineered nanomaterials in the (urban) environment. |

|
Anne Kahru
Anne Kahru is head of the Laboratory of Environmental Toxicology at the National Institute of Chemical Physics and Biophysics, Tallinn, Estonia. Her current research focuses on the mechanisms of (eco) toxicicity and bioavailability of synthetic nanoparticles by combining molecular techniques, in vitro and ecotoxicological tests and analytical chemistry. She is also a founder and President of the Estonian Society of Toxicology. |

|
Sijin Liu
Dr. Sijin Liu is currently a Professor at the Research Center for Eco-Environmental Sciences, the Chinese Academy of Sciences. His research interests interests include: (1) the mechanisms responsible for environmental pollutant-mediated oncogenic effects; (2) nanosafety and nanoimpact. |

|
Willie Peijnenburg
Willie Peijnenburg is Professor of Environmental Toxicology and Biodiversity at Leiden University, The Netherlands. Currently, his main research interests include assessment of the fate and ecological effects of manusfactured nanomaterials in aquatic media. |

|
Debora Rodrigues
Debora Rodrigues is currently an Associate Professor at the University of Houston in the Department of Civil and Environmental Engineering. Her research interests involve investigation of the toxicological effects of carbon-based nanomaterials and polymer nanocomposites to wastewater microbial communities and their potential applications for water treatment and corrosion prevention. |
|
Tara Sabo-Attwood
Tara Sabo-Attwood, PhD is an Associate Professor and Chair of the Department of Environmental and Global Heath, College of Public Health and Health Professions and Center of Environmental and Human Toxicology at the University of Florida. She has broad expertise in environmental molecular toxicology with an emphasis on water and airborne contaminants. |

|
Navid Saleh
Navid Saleh is an Assistant Professor of Civil, Architectural and Environmental Engineering at the University of Texas at Austin. His research focuses on design and development of novel and human-centered water treatment technologies. The primary goal is to enhance economically challenged communities’ access to potable water via innovative nanomaterial-enabled treatment processes. |

|
Gabriele Schaumann
Gabriele Schaumann is Professor of Environmental and Soil Chemistry at the University of Koblenz-Landau, Germany. Her main research interest is to gain a process-orientatied understanding on the fate, transformations and effects of new particulate stressors like engineered nanoparticles and plastic particles in the environment and to adapt and further develop analytical techniques for their detection and characterization in environmental samples. |

|
Vera Slaveykova
Dr. Vera I. Slaveykova is a professor of environmental biogeochemistry and ecotoxicology at the University of Geneva and director of the Department F.-A. Forel for environmental and aquatic sciences at the University of Geneva. Her primary research interests are in development of new tools and concepts to study the basic processes governing the behavior of trace elements and nanoparticles, their interactions with various biotic and abiotic constituents of the aquatic systems, that are highly relevant to water quality and environmental risk assessment. |
 |
Nathalie Tufenkji
Nathalie Tufenkji is Professor in the Department of Chemical Engineering at McGill University, Canada. Her research interests are in the environmental fate of nanomaterials and the development of nano-enhanced products for environmental and biomedical applications (photo credit Eva Blue). |
 |
Maria Elena Vela
Dr. María Elena Vela is a Professor of La Plata National University (Argentina) and researcher at INIFTA, the Research Institute of Theoretical and Applied Physical Chemistry in La Plata city. Her research group works in the synthesis and investigation of functional nanostructured materials and their applications to modify surface properties and to design platforms for ultrasensitive detection of molecules. She also is interested in the study of the interaction of molecules and nanoparticles with model biomembranes.
|
 |
Sharon Walker
Sharon Walker is Interim Dean of UC Riverside’s Bourns College of Engineering, where she also serves as John Babbage Chair in Environmental Engineering and Professor of the Department of Chemical and Environmental Engineering. She is a leading expert on water quality, focusing on the fate and transport of bacteria and nanoparticles in water. |

|
Wendel Wohlleben
Wendel Wohlleben is a Senior Scientist for characterization of nanomaterials at BASF, Dept. of Material Physics. He leads research projects on advanced materials development and on the safety of nanomaterials and is a visiting scientist at the Harvard School of Public Health and at the Department of Materials and Interfaces at the Weizmann Institute, Israel. |
Read some of the high-impact research authored by our new Advisory Board members in Environmental Science: Nano using the links below.
Modeling nanomaterial fate and uptake in the environment: current knowledge and future trends
M. Baalousha, G. Cornelis, T. A. J. Kuhlbusch, I. Lynch, C. Nickel, W. Peijnenburg and N. W. van den Brink
Environ. Sci.: Nano, 2016, 3, 323-345
DOI: 10.1039/C5EN00207A
Effect of humic acid on the kinetics of silver nanoparticle sulfidation
Basilius Thalmann, Andreas Voegelin, Eberhard Morgenroth and Ralf Kaegi
Environ. Sci.: Nano, 2016,3, 203-212
DOI: 10.1039/C5EN00209E
Toxicity of 12 metal-based nanoparticles to algae, bacteria and protozoa
Villem Aruoja, Suman Pokhrel, Mariliis Sihtmäe, Monika Mortimer, Lutz Mädler and Anne Kahru
Environ. Sci.: Nano, 2015,2, 630-644
DOI: 10.1039/C5EN00057B
Toxicity of dimercaptosuccinate-coated and un-functionalized magnetic iron oxide nanoparticles towards aquatic organisms
Ya-Qi Zhang, Ralf Dringen, Charlotte Petters, Wiebke Rastedt, Jan Köser, Juliane Filser and Stefan Stolte
Environ. Sci.: Nano, 2016,3, 754-767
DOI: 10.1039/C5EN00222B
Impact of chemical composition of ecotoxicological test media on the stability and aggregation status of silver nanoparticles
George Metreveli, Bianca Frombold, Frank Seitz, Alexandra Grün, Allan Philippe, Ricki R. Rosenfeldt, Mirco Bundschuh, Ralf Schulz, Werner Manz and Gabriele E. Schaumann
Environ. Sci.: Nano, 2016,3, 418-433
DOI: 10.1039/C5EN00152H
Interaction between palladium-doped zerovalent iron nanoparticles and biofilm in granular porous media: characterization, transport and viability
Mohan Basnet, Alexander Gershanov, Kevin J. Wilkinson, Subhasis Ghoshal and Nathalie Tufenkji
Environ. Sci.: Nano, 2016,3, 127-137
DOI: 10.1039/C5EN00109A
Oral bioavailability and sex specific tissue partitioning of quantum dots in fathead minnows, Pimephales promelas
C. M. Lavelle, J. H. Bisesi, M. A. Hahn, K. J. Kroll, T. Sabo-Attwood and N. D. Denslow
Journal Article Environ. Sci.: Nano, 2015,2, 583-593
DOI: 10.1039/C5EN00122F
End-of-life thermal decomposition of nano-enabled polymers: effect of nanofiller loading and polymer matrix on by-products
Dilpreet Singh, Georgios A. Sotiriou, Fang Zhang, Joey Mead, Dhimiter Bello, Wendel Wohlleben and Philip Demokritou
Environ. Sci.: Nano, 2016, Advance Article
DOI: 10.1039/C6EN00252H
Aerosol synthesis of phase-controlled iron–graphene nanohybrids through FeOOH nanorod intermediates
X. S. Lv, Y. Qiu, Z. Y. Wang, G. M. Jiang, Y. T. Chen, X. H. Xu and R. H. Hurt
Environ. Sci.: Nano, 2016,3, 1215-1221
DOI: 10.1039/C6EN00178E
Surface engineering superparamagnetic nanoparticles for aqueous applications: design and characterization of tailored organic bilayers
Wenlu Li, Carl H. Hinton, Seung Soo Lee, Jiewei Wu and John D. Fortner
Environ. Sci.: Nano, 2016,3, 85-93
DOI: 10.1039/C5EN00089K
Research strategy to determine when novel nanohybrids pose unique environmental risks
Navid B. Saleh, Nirupam Aich, Jaime Plazas-Tuttle, Jamie R. Lead and Gregory V. Lowry
Environ. Sci.: Nano, 2015,2, 11-18
DOI: 10.1039/C4EN00104D
Comments Off on New Advisory Board members for Environmental Science: Nano