In November 2014, the Sustainable Nanotechnology Organization (SNO), a non-profit, international, professional society, held its 3rd annual conference in Boston with over 220 participants in attendance. Drs. Jackie Isaacs of Northeastern University and Philp Demokritou of Harvard University co-chaired the meeting. SNO is dedicated to advancing sustainable nanotechnology around the world through education, research, and responsible growth of nanotechnology.
This themed collection is the summary of representative research papers presented at the Boston conference. Seven eminent scientists and engineers in the field of sustainable nanotechnology gave plenary lectures attended by participants from almost every U.S. state as well as many other countries. About 45% of participants were students, indicative of the recentness of the field.
Selected papers from the conference highlight how sustainable nanotechnology is leading the way to address economic development, global food supplies, as well as energy and water challenges while leaving minimal footprints that can give rise to environmental degradation.
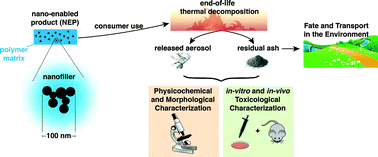
Some of the papers represent the core aspects of sustainable nanotechnology, including biomedical applications, water treatment, green synthesis, life cycle assessments (LCA) and NanoEHS issues. Demokritou et al. present an integrated methodology for the assessment of environmental health implications during thermal decomposition of nano-enabled products.
Demokritou et al., DOI: 10.1039/C4EN00210E
An article by Vicki Grassian et al. reports an important finding that simple nanoscale materials can be complex when considering NanoEHS implications. A number of the fundamental research areas to address NanoEHS needs are suggested.
Grassian et al., DOI: 10.1039/C5EN00112A
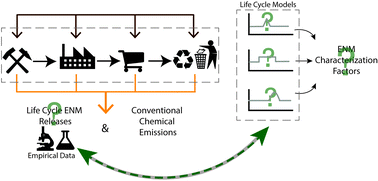
In a review article by Gilbertson, Wender, Zimmerman, and co-workers, the authors summarize recent advances in human and aquatic ecotoxicity life cycle impact assessment for engineered nanomaterials (ENMs) and call for greater coordination between LCA modelers and experimentalists, including those who study fate and transport, environmental transformations, occupational exposure, and toxicology, to inform responsible development of nanotechnology, enabling the technology to reach its full potential.
Gilbertson et al., DOI: 10.1039/C5EN00097A
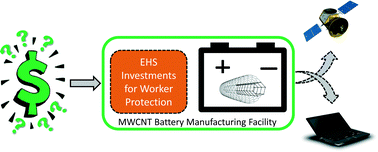
The development of nanomaterials and nano-enabled products in a “greener” manner will minimize any EHS implications while maximizing the societal benefits. Companies working with engineered nanomaterials are expected to make tradeoffs on the costs associated with increased levels of occupational safety and potential environmental impacts. For example, Isaacs et al. present a paper on the economic analysis of carbon nanotube (CNT) lithium-ion battery manufacturing. These authors present a stochastic process-based cost model to investigate the cost drivers for the manufacture of multi-walled CNT nickel manganese cobalt batteries that are targeted for satellite and computer applications. Among other things their results underscore the need for safer manufacturing practices for CNT lithium-ion batteries for application in low and high production volume products such as satellites and portable computers, respectively.
Isaacs et al., DOI: 10.1039/C5EN00078E
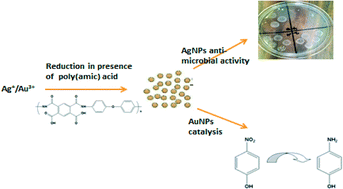
Greener nanotechnology can be the “role model” for industrial development in the 21st century. Sadik et al. demonstrate that a one-pot synthesis of silver and gold nanoparticles is possible using conductive, electroactive, and biodegradable polymers. In addition to modest cytotoxicity against non-cancerous, immortalized and cancerous cell lines, the synthesized nanoparticles exhibit excellent antibacterial activity against gram negative and gram positive bacteria.
Sadik et al., DOI: 10.1039/C5EN00053J
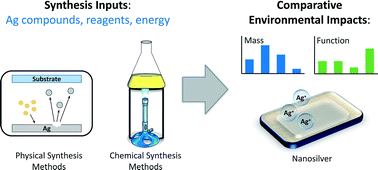
Pourzahedi et al. apply green chemistry and sustainable manufacturing to nanomaterial synthesis, with the goal of reducing life cycle energy use and environmental impacts. The authors use LCA to analyze and compare the environmental impacts of AgNPs produced through seven different synthesis routes (cradle-to-gate). LCA reveals both direct and indirect or upstream impacts associated with AgNPs. Results show that across synthesis routes, impacts associated with the upstream production of bulk silver itself are dominant for nearly every category of environmental impact, contributing to over 90% of life cycle burdens in some cases. The bio-based chemical reduction route has important tradeoffs in ozone depletion potential and ecotoxicity.
Pourzahedi et al., DOI: 10.1039/C5EN00075K
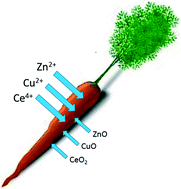
The release of ENMs into the environment has led to concerns about the potential risks to food safety and human health. Ebbs et al. describe the extent of ENM uptake into plant foods. The authors focus on the accumulation of zinc, copper, or cerium in carrot exposed to metal oxide nanoparticles and metal ions. They demonstrate that ENMs are no more toxic than the ionic treatments and show a reduced accumulation in the edible tissues of carrot. The results demonstrate that the understanding of ionic metal transport in plants may not accurately predict ENM transport and that an additional comparative study is needed for this and other crop plants.
Ebbs et al., DOI: 10.1039/C5EN00161G
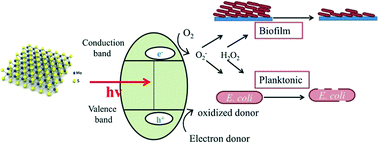
Rodrigues et al. provide an assessment of the toxicity of exfoliated-MoS2 and annealed exfoliated-MoS2 towards planktonic cells, biofilms, and mammalian cells in the presence of electron donor.
Rodrigues et al., DOI: 10.1039/C5EN00031A
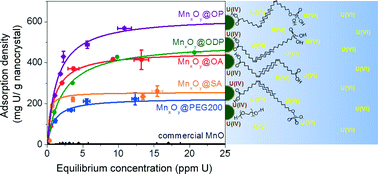
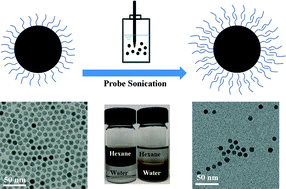
Lee et al. report the development of precisely engineered manganese oxide nanoscale particles for the sorption of uranium as uranyl in water. They synthesize nanoparticles via thermal decomposition of manganese oleate and then phase-transfer the particles into water using ligand exchange and bilayer stabilization methods. The resulting monodisperse suspensions demonstrate significantly enhanced uranyl adsorption as a function of size, surface coating chemistries, and solution pH.
Lee et al., DOI: 10.1039/C5EN00010F
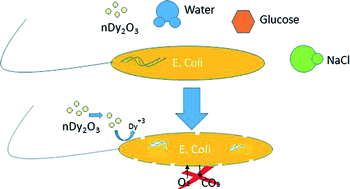
The fate of dysprosium oxide nanoparticles (Dy2O3) and their effects on natural biological systems are a growing concern. Oyanedel-Craver et al. have assessed the toxicity of nDy2O3 on Escherichia coli for concentrations between 0.02 and 2 mg/L exposed to three concentrations of NaCl and three glucose concentrations. Toxicity measurement of Dysprosium ion Dy(+3) suggest that it is the main contributor to the overall toxicity.
Oyanedel-Craver et al., DOI: 10.1039/C5EN00074B
Among other applications, engineered superparamagnetic nanoparticles have broad potential in biotechnologies, high contrast magnetic resonance imaging, and advanced environmental sensing and remediation technologies. Fortner et al. present the flexible surface design strategies for a variety of superparamagnetic iron oxide nanoparticles for applications in aqueous systems.
Fortner et al., DOI: 10.1039/C5EN00089K
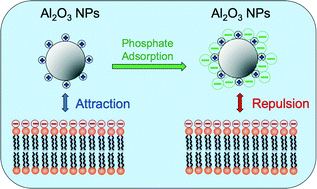
Chen et al. describe the aggregation and interactions of chemical mechanical planarization nanoparticles with model biological membranes, focusing on the role of phosphate adsorption.
Chen et al., DOI: 10.1039/C5EN00176E
The difficulty of meeting the world’s energy demand is compounded by the growing need to protect human health and the environment. Nanotechnology will play a major role in the development of clean, affordable, and renewable energy sources. Soroush et al. demonstrate that silver nanoparticle (AgNPs)-decorated graphene oxide (GO) functionalized membranes exhibit super-hydrophilic properties with contact angles below 25°. The membranes also exhibit significant E. coli inactivation without adversely affecting the membrane transport properties. Such membrane could be employed as composites of forward osmosis and seawater desalination because of its energy efficiency.
Soroush et al., DOI: 10.1039/C5EN00086F
We hope you enjoy this issue which represents a snapshot of the wider conversation on the topic of sustainable nanotechnology. We also invite you to visit us at www.susnano.org as we develop a framework for using nanotechnology to address grand global challenges in the energy, water, and food sectors while maintaining a balance between the economic, environmental, and societal issues.
Enjoy this issue!
Wunmi Sadik, President & Co-founder
Barbara Karn, Executive Director & Co-founder
Jacqueline Isaacs and Philip Demokritou, SNO 2014 Co-Chairs