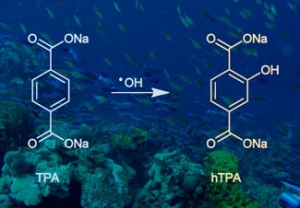
Vera Thoss tells Elinor Richards about her bluebell business and research, using her car in her experiments and analysing whale vomit
 Vera Thoss is an environmental chemistry lecturer at Bangor University, UK. Her research is based on ecological chemistry, which addresses processes mediated through specific compounds within ecosystems and environmental chemistry, which is concerned with the impact of human activities on the environment.
Vera Thoss is an environmental chemistry lecturer at Bangor University, UK. Her research is based on ecological chemistry, which addresses processes mediated through specific compounds within ecosystems and environmental chemistry, which is concerned with the impact of human activities on the environment.
What inspired you to become a scientist?
It all started when I was 13 and I had my first chemistry lesson. I instantly took to the subject and from then my mind was made up. I was also curious and wanted to ‘understand the world’.
What attracted you to environmental science?
As a chemist, the choice was between synthetic and analytical chemistry. I chose analytical chemistry because it allows you to follow the environmental fate of natural or man-made compounds. Being allowed to spend time in the woods was a big bonus!
What projects are you working on?
Currently, my group is working on oil pollution, composting and plant-derived products. It seems a bit of a stretch but it is all part of carbon cycling: plants build precious molecules, most of the time these remain intact but may transfer into air, water or soil. Crude oil is the remnants of sunken forests. So in the end, all the chemistry comes from plants photosynthesising and creating complex fragrances, tastes and colours. It is fascinating.
What will be the next big breakthrough in your field?
To chemically separate plant material into multiple useable compounds, with environmentally benign techniques, using as little energy as possible and ideally producing no waste at all.
Which achievements are you most proud of?
My beautiful daughter. Last year I organised the first ‘Plants as Providers of Fine Chemicals’ conference, which was very successful. I also managed to measure picogram amounts of monoterpenes in three-week-old Scots pine seedlings before they were eaten by slugs.
You own a farm from which you run a business selling bluebells called Vera Bluebell. How did this come about?
I was always concerned about the availability of clean drinking water, and moving to a mountain farm near Snowdon in Wales was a strategic choice (it does rain a lot!). Realising that there was abundance of bluebells on the land was a chance discovery after a fire. I was aware of their unusual chemistry though and that was the starting point for Vera Bluebell. Bluebells are protected, which means a license is needed to work with them. There was a demand for wild bluebells as well. We have now been sustainably managing a wild bluebell population for over six years and it has been an interesting journey. I would love to see a bluebell derived extract being used in a commercial product.
What discoveries have you made during your research on bluebells?
Bluebell seeds have a high oil content and the oil has an unusual composition. Even though this is the first chemical assessment of Hyacinthoides non-scripta oil, the chemistry is not earth-shattering. The ecology aspects gave room for more discoveries, for example we found seed stores on the site, meaning that possibly voles or shrew have collected the seeds for storage. This has never been reported before.
Tell us about your bluebell conservation efforts and how your research can help.
We are hoping to show that bluebell seeds can be a source of fine chemicals. We obtain an oil of unusual composition from the seeds. The residue contains iminosugars, which may be of use in future medicines. I am hoping that the compounds isolated from bluebell seeds will be of commercial value, which in turn means that the conservation of bluebells pays for itself. We are hoping to paint the mountains and woodlands blue again.
You’re involved with projects called BEACON and PROBECO. What are these and what is your role in both projects?
BEACON is all about biorefining, obtaining different compounds from the same plant feedstock. There are different feedstocks investigated in BEACON ranging from perennial rye grass to ivy. My role is to analyse whole plant composition and organise the ‘ Plants as Providers of Fine Chemicals ‘ conference. The PROBECO project was about the influence of monoterpenes on ecosystem processes in Caledonian Scots pine forests. These are very rare ecosystems. Individual pine trees smell different and the forest served as a study site to investigate the role of specific monoterpenes. I was the scientist analysing the smell of thousands of pine trees and we came up with the chemodiversity hypothesis.
In 2007, you worked with Welsh company Used Tyre Distillation Research to produce novel products from used tyres, in particular oil for fuelling cars. What was your role in the research? I read that your car was used to test the fuel. What was the result?
Again my role was to analyse the products. The oil was a complex mixture and we did see some interesting compounds in it. The tyre oil was noticeably energy dense, giving faster acceleration to the car, which has survived the experiment well!
In 2008, you had an odd request to analyse what was thought to be whale vomit (ambergris) on a North Wales beach. Why is whale vomit so important and what did you find?
I’ve had a few requests for the analysis of beach finds. Ambergris is sought after in the perfume industry, but we have yet to get our hands on some. Most samples were waxes or plastic, which possibly fell overboard, just aged in the sea.
What other odd things have you been asked to analyse?
Another nice in-house example for analysis was to trace a smell in the corridor back to its origin: we sampled air in the corridor outside my office and the terrible smell was found to be due to demolition work going on next door. If it stinks don’t automatically blame the chemists!
What do you do in your spare time?
I love gardening, farming and generally just being outdoors. I am an amateur bee keeper and enjoy spending time with my family.
Read the original article at Chemistry World, or Vera’s recent paper in the journal RSC Advances:
Triacylglycerol composition of British bluebell (Hyacinthoides non-scripta) seed oil
Vera Thoss, P J Murphy, Ray John Marriott and Thomas Wilson
DOI: 10.1039/C2RA20090B











 Gregory Korshin talks to Michael Smith about his passion for environmental science, literature and languages
Gregory Korshin talks to Michael Smith about his passion for environmental science, literature and languages