Probably not the type of diet you are thinking of. It has something to do with food, though. I am talking about the transfer mechanisms some bacteria use to share metabolites between them, and also with bacteria of different species. In this review published in Environmental Science: Processes & Impacts, two researchers from North Carolina analyse the latest discoveries in this field, specifically in mechanisms known as DIET: “direct interspecies electron transfer”.
Scientists discovered DIET not a decade ago. Before that, only conventional diffusion models were known: an organism generated an excess of certain metabolite, released it to the surrounding media and was consumed by a second organism. This is called “mediated interspecies electron transfer” (MIET).
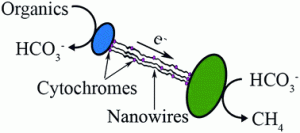
DIET, however, is more efficient. Instead of releasing the metabolites around, bacteria use structures that allow them to transfer chemical substances directly to other cells. These structures, usually filamentous pili full of conductive cytochromes, act as nanowires that connect bacteria to one another. However, because DIET is a form of electron transfer, sometimes proteins are not needed at all and actual electric cables may be used. When pili and cytochromes are removed in genetically engineered bacteria, they can use metal, metal oxides (such as magnetite) or activated carbon as connections. Sometimes bacteria prefer this cables to their own traditional methods: in some experiments, scientists showed that bacteria will rather connect to carbon than to other cells, probably due to the higher electric conductivity.
Nowadays we use multicellular bacterial communities in a wide variety of industrial systems from sewage treatment to energy production. Understanding how DIET interactions work is key to improve the effectiveness of these processes and will allow us to have a better control. Who knows, maybe some day DIET will lead into building intelligent bacterial circuits, the same way years ago silicon allowed us to create microchips.
Read the Critical Review for free* today:
Hardwiring microbes via direct interspecies electron transfer: mechanisms and applications
Qiwen Cheng and Douglas F. Call
Environ. Sci.: Processes Impacts, 2016,18, 968-980
DOI: 10.1039/C6EM00219F
*Access is free until 23/09/2016 through a registered publishing personal account.