Heavy metals are toxic, but sometimes their organometallic compounds are worse. In this respect, methylmercury (MeHg+) represents an excellent example: the detrimental health and environmental effects associated with mercury (Hg) pollution are in fact caused by MeHg+, and not by the metal itself. Methylmercury is produced in the aquatic environment by archea and anoxic bacteria, which use inorganic mercury as a starting substrate. Once produced, MeHg+ enters the food-web and accumulates in organisms such as fish and seafood, which represents the main methylmercury exposure pathway for the human beings.
Due to its high neurotoxicity, many efforts have been devoted to improve our knowledge on the environmental fate of methylmercury. In this context, identify and characterize MeHg+ degradation mechanisms represent an important step, as the degradation processes will directly affect methylmercury concentrations in the environment, thus the dose at which we are exposed.
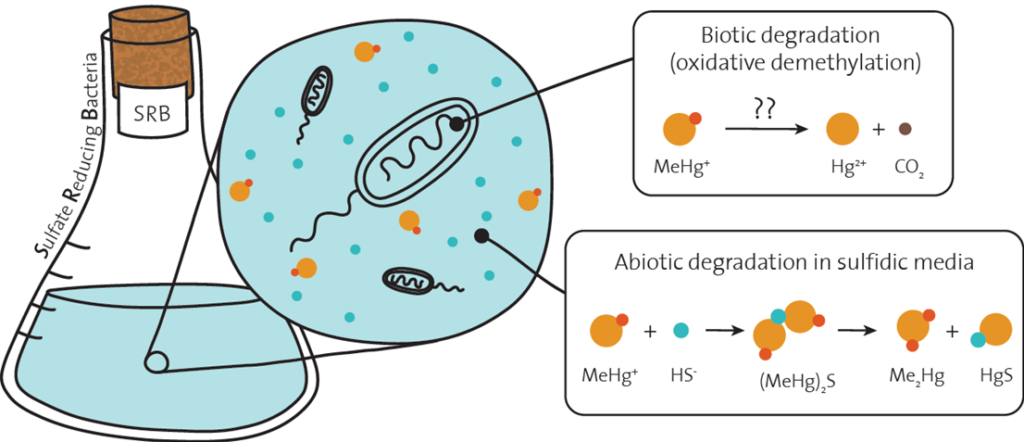
So far, four different MeHg+ degradation pathways have been identified. The so-called oxidative demethylation involves the degradation of MeHg+ to inorganic mercury and carbon dioxide by anaerobic bacteria. Differently from the other three processes, which are relatively well understood, many doubts still remain on the details of this pathway despite almost 30 years of scientific investigations. So many, in fact, to bring some researchers back to the literature with a big question to be answered: is oxidative demethylation a real process, or it is just an experimental artifact?
Based on the available literature on methylmercury chemistry and biogeochemistry, Kanzler et al. hypothesized that a simple abiotic process could be responsible for the MeHg+ degradation that have been observed in the presence of sulfate-reducing bacteria, the microorganisms that are believed to perform oxidative demethylation. The proposed reaction involves the formation of a binuclear complex, bis(methylmercury) sulphide ((MeHg)2S), which can degrade to insoluble cinnabar (HgS) and dimethylmercury (Me2Hg), a volatile compound.
2MeHg+ + HS– ⇌ (MeHg)2 S + H+→ Me2Hg + HgS
Several factors influence the thermodynamics and the kinetics of this reaction, including the solution pH and the concentrations of the two precursors, methylmercury and hydrogen sulfide. Using a combination of experimental and computational approaches, Kanzler et al. developed a model that allows the prediction of both MeHg+ speciation (i.e., the position of the first equilibrium) and dimethylmercury formation rates as a function of the chemical composition of the medium.
Based on this model, the authors showed that the binuclear complex bis(methylmercury) sulphide is the dominant methylmercury species in the pure culture studies that previously investigated the oxidative demethylation pathway. In other words, the methylmercury loss was most likely associated to the abiotic formation of (MeHg)2S, rather than to an enzymatic degradation process. Notably, these former microbiological studies lack abiotic control experiments that might undoubtedly prove or disprove Kanzler’s conclusions.
The model was also employed to investigate whether (MeHg)2S decomposition might be a relevant dimethylmercury (Me2Hg) formation pathway in the natural environment. Dimethylmercury represents almost half of the total mercury species in the open ocean, but so far very few explanations have been proposed regarding its formation mechanism. Despite being an appealing hypothesis, very slow Me2Hg formation rates were anticipated in environmentally-relevant conditions, implying that (MeHg)2S decomposition can be a relevant dimethylmercury formation process only in environments with long MeHg+ residence times, i.e. in the subsurface ocean.
In this study, Kanzler et al. cast new lights on aspects of the biogeochemical cycle of MeHg+ that are still poorly understood. New work is now needed to confirm the model’s prediction and to update our knowledge on the environmental fate of this neurotoxic compound.
To download the full article for free* click the link below:
Emerging investigator series: methylmercury speciation and dimethylmercury production in sulfidic solutions
Charlotte R. Kanzler, Peng Lian, Emma Leverich Trainer, Xiaoxuan Yang, Niranjan Govind, Jerry M. Parks and Andrew M. Graham
Environ. Sci. Process. Impacts, 2018, 20, 584–594
DOI: 10.1039/C7EM00533D
About the Webwriter:
Rachele Ossola is a PhD student in the Environmental Chemistry group at ETH Zurich. Her research focuses on photochemistry of dissolved organic matter in the natural environment.
*Free to read until 31st August 2018