The chemical behaviour of pollutants in the aquatic environment requires careful monitoring in order for us to understand the toxicity different compounds will exert on natural ecosystems. Researchers from Israel describe a new modelling approach that may provide an exciting new technique to address a specific aspect of the chemical and environmental behaviour of pollutants.
The term ‘chiral’ is given to molecules that exhibit identical composition, but where the components of the molecule are arranged in a non-superimposable mirror image composition, centred around an asymmetric carbon atom. The two ‘mirror images’ of a chiral molecule are termed enantiomers. The study of the enantiomer-specific properties and how these vary between different molecules is of major interest within the broad fields of inorganic, organic, physical, biological and environmental chemistry.
Many anthropogenic chemicals of environmental concern, such as pesticides, are chiral molecules. These compounds can potentially be major threat to aquatic ecosystems. For example, the molecular structures and environmental implications of many chiral pesticides have been discussed in a review by the USEPA. It is important, therefore, to have means of accurately tracing the alteration of these compounds in the environment, particularly with reference to their enantiomer-specific environmental toxicity.
Researchers have previously proposed an enantiomeric enrichment factor (EEF), to describe the enantiomeric enrichment – conversion relationship of chiral compounds, derived using the Rayleigh equation, which describes the relationship between changes in the isotopic composition against the contaminant concentration during the degradation process. The EEF can therefore be used as an identifying tool for a specific enzymatic reaction of different molecules.
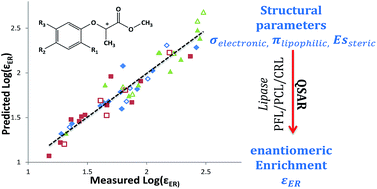
Developing models to describe enantio-selective biodegradation can alleviate the need for laborious practical work. To achieve this, there is a need to improve our understanding of the mechanisms of biodegradation, to classify chemicals according to their relative biodegradability, and to develop reliable biodegradation estimation methods for new chemicals. Quantitative structure–activity relationship (QSAR) models are typically derived based on the correlation between experimental data and physical properties (e.g. lipophilicity, steric and electronic parameters) and can be used to identify bioavailability, toxicity and biological activities of compounds as dependent variables.
This study, conducted by researchers from The Institute of Chemistry at The Hebrew University of Jerusalem and The Geological Survey of Israel, develops a QSAR model to describe the dependence of the enantiomeric enrichment factor on molecular structures and uses this method to evaluate EEF values for unstudied chiral compounds. The authors used the multiple linear regression (MLR) method to build the QSAR based on the Linear Hansch model. The enantioselective hydrolysis of 16 derivatives of 2-(phenoxy)propionate (PPMs) (some of which are common herbicides) using three different lipase enzymes was analysed.
The study provides a demonstration of the predictive power of QSAR and Hansch modeling for analysis of the structural dependence of the EEF, with the model shown to effectively correlate biological activity with key physicochemical properties. More importantly, at times, the QSAR of EEF values was shown to be a much better predictive tool than the QSAR of just the underlying individual kinetic parameters, clearly indicating this method could mark the way forward for research in this field.
The authors note that the use of the QSAR modelling technique used in this study may serve as a powerful tracer tool in environmental studies, assisting in source tracking the enantio-selective conversion of both known and unstudied chiral compounds in aquatic ecosystems.
To read more about this study, download a copy for free* by clicking the link below.
Quantitative structure–activity relationship correlation between molecular structure and the Rayleigh enantiomeric enrichment factor
S. Jammer, D. Rizkov, F. Gelman and O. Lev
Environ. Sci.: Processes Impacts, 2015, Advance Article
DOI: 10.1039/c5em00084j
—————-
Ian Keyte is a Doctoral Researcher at the University of Birmingham. His research focuses on the sources, behavior and fate of polycyclic aromatic hydrocarbons (PAHs) in the atmosphere.
—————-
* Access is free until 30/08/2015 through a registered RSC account.