Abigail Hallowes writes about an EES article in Chemistry World
Researchers from the US have optimised a photoelectrochemical cell (PEC) so that it can continuously split water into clean burning hydrogen and oxygen for over 2200 hours – the equivalent to one year of outdoor operation.
In order for PECs to be a competitive energy provider, they should efficiently covert solar into chemical energy, whilst remaining stable for years of continuous operation. The condition of the electrodes in PECs is a major concern as they immediately start to corrode once immersed in the electrolyte. Previous PEC researchers have only been able to produce systems that are either stable for 4–100 hours but inefficient, or efficient but only stable for a few minutes; they have not been able to incorporate both required properties.

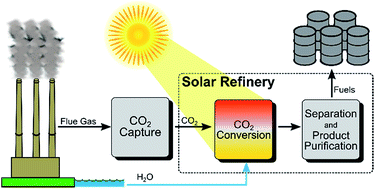
Scanning-electron micrograph image of a microwire array with its protective coating
Electrode instability is brought on when the energy required to excite an electron is within the same energy range that causes electrode corrosion; photocorrosion then becomes competitive with water splitting. In these cases, it is most likely that photocorrosion is thermodynamically more favourable which therefore leads to unstable electrodes. Now, a team led by Nathan Lewis at the California Institute of Technology have drastically increased PEC stability whilst achieving a 100% Faradaic efficiency for oxygen evolution.
Their approach increased the electrochemically active sites versus the surface area of the electrode through the use of silicon microwire arrays; this led to a decrease in the effective current density at the electrode–electrolyte interface, increasing the energy required for photocorrosion, which decreased the rate of photocorrosion. They also coated the arrays in a protective but conductive layer, to act as a corrosion-resistant barrier while maintaining efficient charge transfer to the reaction sites, as well as with an oxygen evolution catalyst to promote water oxidation.
Brian Seger, a photoelectrochemist at the Technical University of Denmark, explains that water splitting provides some very corrosive conditions so stability has been a large hurdle in this field: ‘The fact that the Lewis group could test their device for three months with no noticeable corrosion indicates that this hurdle is surmountable.’
Materials scientist, Dongyuan Zhao, of Fudan University, China, describes the work was a breakthrough and says it ‘shows great potential for industrial application.’
Interested to find out more? Read the full article by Abigail Hallowes in Chemistry World.
Read the original article in Energy and Environmental Science:
Stabilization of Si microwire arrays for solar-driven H2O oxidation to O2(g) in 1.0 M KOH(aq) using conformal coatings of amorphous TiO2
Matthew R. Shaner,ab Shu Hu,ab Ke Sunab and Nathan S. Lewis
Energy Environ. Sci., 2015, Advance Article
DOI: 10.1039/C4EE03012E